plant
life form
Introduction

 any member of the kingdom Plantae, multicellular, eukaryotic life forms characterized by (1) photosynthetic nutrition, in which chemical energy is produced from water, minerals, and carbon dioxide with the aid of pigments and the radiant energy of the Sun, (2) essentially unlimited growth at localized regions, (3) cells that contain cellulose in their walls and are therefore to some extent rigid, (4) the absence of organs of locomotion, resulting in a more or less stationary existence, (5) the absence of sensory and nervous systems, and (6) life histories that show an alteration of haploid and diploid generations, with the dominance of one over the other being taxonomically significant.
any member of the kingdom Plantae, multicellular, eukaryotic life forms characterized by (1) photosynthetic nutrition, in which chemical energy is produced from water, minerals, and carbon dioxide with the aid of pigments and the radiant energy of the Sun, (2) essentially unlimited growth at localized regions, (3) cells that contain cellulose in their walls and are therefore to some extent rigid, (4) the absence of organs of locomotion, resulting in a more or less stationary existence, (5) the absence of sensory and nervous systems, and (6) life histories that show an alteration of haploid and diploid generations, with the dominance of one over the other being taxonomically significant.
 Plants range in size from diminutive duckweeds only a few millimetres in length to the giant sequoias of California that reach 90 metres (300 feet) or more in height. There are approximately 275,000 to 300,000 different species of plants, and new species are continuously being described, particularly from unexplored tropical areas of the world. Having evolved from aquatic ancestors, plants have subsequently migrated over the entire surface of the Earth, inhabiting tropical, Arctic, desert, and Alpine regions. Some plants have returned to an aquatic habitat in either fresh or salt water.
Plants range in size from diminutive duckweeds only a few millimetres in length to the giant sequoias of California that reach 90 metres (300 feet) or more in height. There are approximately 275,000 to 300,000 different species of plants, and new species are continuously being described, particularly from unexplored tropical areas of the world. Having evolved from aquatic ancestors, plants have subsequently migrated over the entire surface of the Earth, inhabiting tropical, Arctic, desert, and Alpine regions. Some plants have returned to an aquatic habitat in either fresh or salt water.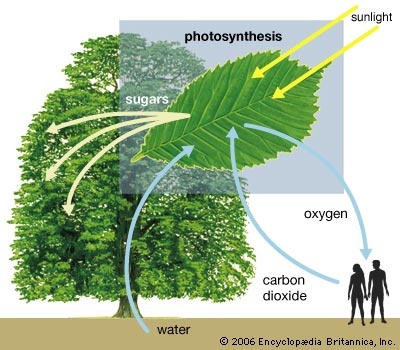 Plants play a vital role in the maintenance of life on Earth. All energy used by living organisms depends on the complex process of photosynthesis, which is carried out by green plants. Radiant energy from the Sun is transformed into organic chemical energy in the form of sugar through the fundamental series of chemical reactions constituting photosynthesis. In nature all food chains begin with green plants (primary producers). Primary producers, represented by trees, shrubs, and herbs, are a prolific source of energy in the form of carbohydrates (sugars) stored in the leaves. These carbohydrates, produced in photosynthesis, are broken down in a process called respiration; the smaller units of the sugar molecule and its products fuel numerous metabolic processes. Various parts of the plant (e.g., leaves) are the energy sources that support animal life in different community habitats. A by-product of photosynthesis, oxygen, is essential to animals.
Plants play a vital role in the maintenance of life on Earth. All energy used by living organisms depends on the complex process of photosynthesis, which is carried out by green plants. Radiant energy from the Sun is transformed into organic chemical energy in the form of sugar through the fundamental series of chemical reactions constituting photosynthesis. In nature all food chains begin with green plants (primary producers). Primary producers, represented by trees, shrubs, and herbs, are a prolific source of energy in the form of carbohydrates (sugars) stored in the leaves. These carbohydrates, produced in photosynthesis, are broken down in a process called respiration; the smaller units of the sugar molecule and its products fuel numerous metabolic processes. Various parts of the plant (e.g., leaves) are the energy sources that support animal life in different community habitats. A by-product of photosynthesis, oxygen, is essential to animals. The daily existence of human beings is also directly influenced by plants. Plants furnish food and flavourings; raw materials for industry, such as wood, resins, oils, and rubber; fibres for the manufacture of fabrics and cordage; medicines; insecticides; and fuels. More than half of the Earth's population relies on the grasses rice, corn (maize), and wheat as their primary source of food. Apart from their commercial and aesthetic value, plants conserve other natural resources by protecting soils from erosion, by controlling water levels and quality, and by producing a favourable atmosphere.
The daily existence of human beings is also directly influenced by plants. Plants furnish food and flavourings; raw materials for industry, such as wood, resins, oils, and rubber; fibres for the manufacture of fabrics and cordage; medicines; insecticides; and fuels. More than half of the Earth's population relies on the grasses rice, corn (maize), and wheat as their primary source of food. Apart from their commercial and aesthetic value, plants conserve other natural resources by protecting soils from erosion, by controlling water levels and quality, and by producing a favourable atmosphere.The following article summarizes the morphological, physiological, and ecological features of plants. The principal focus is on structure and function, physiology, life histories, and ecology, and on how the various plant groups have evolved, dispersed, and become adapted to life on land. The features that define each major plant group and the role they play in the wider ecosystem also are discussed.
Definition of the kingdom
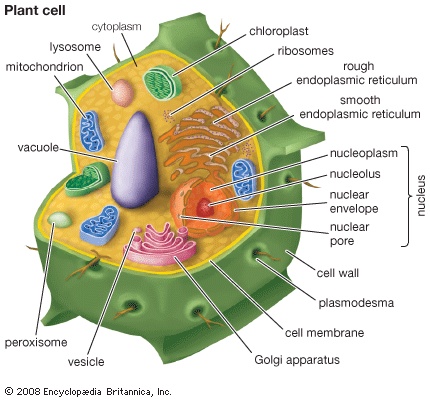 The kingdom Plantae includes organisms that range in size from a tiny moss to a giant tree. Despite this enormous variation, all plants are multicellular and eukaryotic (i.e., each cell possesses a membrane-bound nucleus that contains the chromosomes). They generally possess pigments (chlorophylls a and b and carotenoids), which play a central role in converting the energy of sunlight into chemical energy by means of photosynthesis. Plants, therefore, are independent in their nutritional needs (autotrophic) and store their excess food in the form of macromolecules of starch. The relatively few plants that are not autotrophic have lost pigments and are dependent on other organisms for nutrients (parasitism). Although plants are nonmotile organisms, some produce motile cells (gametes) propelled by whiplike flagella. Plant cells are surrounded by a more or less rigid cell wall composed of the carbohydrate cellulose, and adjacent cells are interconnected by microscopic strands of cytoplasm called plasmodesmata, which traverse the cell walls. Many plants have the capacity for unlimited growth at localized regions of cell division, called meristems. Plants, unlike animals, can use inorganic forms of the element nitrogen (N), such as nitrates and ammonia, and thus do not require an external source of protein (in which nitrogen is a major constituent) to survive.
The kingdom Plantae includes organisms that range in size from a tiny moss to a giant tree. Despite this enormous variation, all plants are multicellular and eukaryotic (i.e., each cell possesses a membrane-bound nucleus that contains the chromosomes). They generally possess pigments (chlorophylls a and b and carotenoids), which play a central role in converting the energy of sunlight into chemical energy by means of photosynthesis. Plants, therefore, are independent in their nutritional needs (autotrophic) and store their excess food in the form of macromolecules of starch. The relatively few plants that are not autotrophic have lost pigments and are dependent on other organisms for nutrients (parasitism). Although plants are nonmotile organisms, some produce motile cells (gametes) propelled by whiplike flagella. Plant cells are surrounded by a more or less rigid cell wall composed of the carbohydrate cellulose, and adjacent cells are interconnected by microscopic strands of cytoplasm called plasmodesmata, which traverse the cell walls. Many plants have the capacity for unlimited growth at localized regions of cell division, called meristems. Plants, unlike animals, can use inorganic forms of the element nitrogen (N), such as nitrates and ammonia, and thus do not require an external source of protein (in which nitrogen is a major constituent) to survive.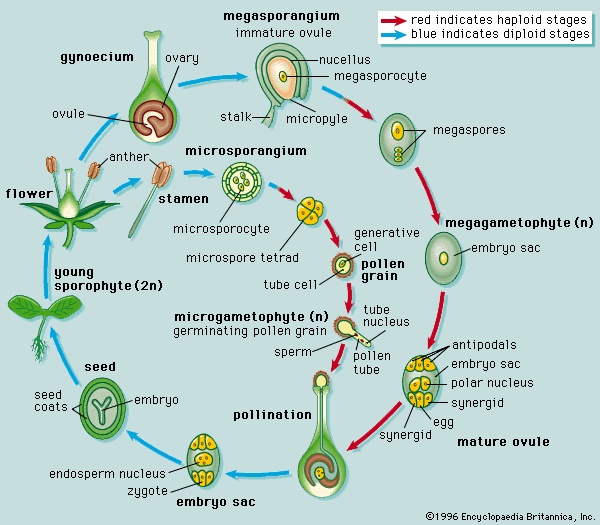 The life histories of plants include two phases, or generations, one of which is diploid (the nuclei of the cells contain two sets of chromosomes), while the other is haploid (with one set of chromosomes). The diploid generation is known as the sporophyte, which literally means spore-producing plant. The haploid generation, called the gametophyte, produces the sex cells, or gametes. The complete life cycle of a plant thus involves an alternation of haploid and diploid generations. The sporophyte and gametophyte generations of plants are structurally quite dissimilar.
The life histories of plants include two phases, or generations, one of which is diploid (the nuclei of the cells contain two sets of chromosomes), while the other is haploid (with one set of chromosomes). The diploid generation is known as the sporophyte, which literally means spore-producing plant. The haploid generation, called the gametophyte, produces the sex cells, or gametes. The complete life cycle of a plant thus involves an alternation of haploid and diploid generations. The sporophyte and gametophyte generations of plants are structurally quite dissimilar. The concept of what constitutes a plant has undergone significant change over time. For example, at one time the photosynthetic aquatic organisms commonly referred to as algae were considered members of the plant kingdom. The various major algal groups, such as the green algae, brown algae, and red algae, are now placed in the kingdom Protista because they lack one or more of the features that are characteristic of plants. The organisms known as fungi (fungus) also were once considered to be plants since they reproduce by spores and possess a cell wall. The fungi, however, uniformly lack chlorophyll (heterotrophic) and are chemically distinct from the plants; they are now placed in a separate kingdom, Fungi.
The concept of what constitutes a plant has undergone significant change over time. For example, at one time the photosynthetic aquatic organisms commonly referred to as algae were considered members of the plant kingdom. The various major algal groups, such as the green algae, brown algae, and red algae, are now placed in the kingdom Protista because they lack one or more of the features that are characteristic of plants. The organisms known as fungi (fungus) also were once considered to be plants since they reproduce by spores and possess a cell wall. The fungi, however, uniformly lack chlorophyll (heterotrophic) and are chemically distinct from the plants; they are now placed in a separate kingdom, Fungi.No definition of the kingdom completely excludes all nonplant organisms or even includes all plants. There are plants, for example, that do not produce their food by photosynthesis but rather are parasitic on other living plants; other plants are saprophytic, obtaining their food from dead organic matter. Some animals possess plantlike characteristics, such as the lack of mobility (e.g., sponges) or the presence of a plantlike growth form (e.g., some corals and bryozoans), but in general such animals lack the other characteristics of plants cited here.
Despite such differences, plants share the following features common to all living things. Their cells undergo complex metabolic reactions that result in the production of chemical energy, nutrients, and new structural components. They respond to internal and external stimuli in a self-preserving manner. They reproduce by passing their genetic information to descendants that resemble them. They have evolved over geologic time by the process of natural selection into a wide array of forms and life-history strategies.
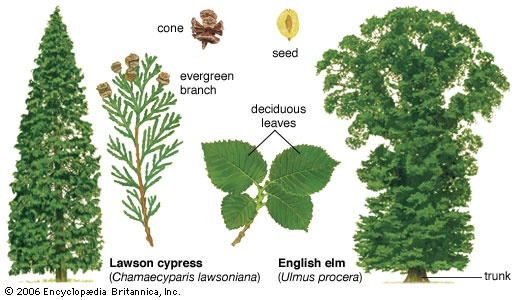
 The earliest plants undoubtedly evolved (evolution) from an aquatic, green algal ancestor (as evidenced by similarities in pigmentation, cell wall chemistry, biochemistry, and method of cell division), and different plant groups have become adapted to terrestrial life to varying degrees. Land plants face severe environmental threats or difficulties, such as desiccation, drastic changes in temperature, support, nutrient availability to each of the cells of the plant, regulation of gas exchange between the plant and the atmosphere, and successful reproduction. Thus, many adaptations to land existence have evolved in the plant kingdom and are reflected among the different major plant groups. An example is the development of a waxy covering (the cuticle) that covers the plant body, preventing excess water loss. Specialized tissues and cells (vascular tissue) enabled early land plants to absorb and transport water and nutrients to distant parts of the body more effectively and, eventually, to develop a more complex body composed of organs called stems, leaves, and roots. The evolution and incorporation of the substance lignin into the cell walls of plants provided strength and support. Details of the life history are often a reflection of a plant's adaptation to a terrestrial mode of life and may characterize a particular group; for example, the most highly evolved plants reproduce by means of seeds, and, in the most advanced of all plants (angiosperms), a reproductive organ called a flower is formed.
The earliest plants undoubtedly evolved (evolution) from an aquatic, green algal ancestor (as evidenced by similarities in pigmentation, cell wall chemistry, biochemistry, and method of cell division), and different plant groups have become adapted to terrestrial life to varying degrees. Land plants face severe environmental threats or difficulties, such as desiccation, drastic changes in temperature, support, nutrient availability to each of the cells of the plant, regulation of gas exchange between the plant and the atmosphere, and successful reproduction. Thus, many adaptations to land existence have evolved in the plant kingdom and are reflected among the different major plant groups. An example is the development of a waxy covering (the cuticle) that covers the plant body, preventing excess water loss. Specialized tissues and cells (vascular tissue) enabled early land plants to absorb and transport water and nutrients to distant parts of the body more effectively and, eventually, to develop a more complex body composed of organs called stems, leaves, and roots. The evolution and incorporation of the substance lignin into the cell walls of plants provided strength and support. Details of the life history are often a reflection of a plant's adaptation to a terrestrial mode of life and may characterize a particular group; for example, the most highly evolved plants reproduce by means of seeds, and, in the most advanced of all plants (angiosperms), a reproductive organ called a flower is formed.Nonvascular plants
Definition of the category
 The plant division Bryophyta includes tiny plants that lack specialized vascular tissue (xylem and phloem) for internal water and food conduction and support. Bryophytes (bryophyte) are, therefore, nonvascular plants and, correlatively, possess no true roots, stems, or leaves. Some larger mosses, however, contain a central core of elongated, thick-walled cells called hydroids that are involved in water conduction and that have been compared to the xylem elements of other plants. The Bryophyta are second in diversity among the land plants only to the flowering plants (angiosperms) and are generally regarded as composed of three classes; some evidence indicates, however, that these groups may not be so closely related: Musci (the mosses), Hepaticae (the liverworts), and Anthocerotae (the hornworts).
The plant division Bryophyta includes tiny plants that lack specialized vascular tissue (xylem and phloem) for internal water and food conduction and support. Bryophytes (bryophyte) are, therefore, nonvascular plants and, correlatively, possess no true roots, stems, or leaves. Some larger mosses, however, contain a central core of elongated, thick-walled cells called hydroids that are involved in water conduction and that have been compared to the xylem elements of other plants. The Bryophyta are second in diversity among the land plants only to the flowering plants (angiosperms) and are generally regarded as composed of three classes; some evidence indicates, however, that these groups may not be so closely related: Musci (the mosses), Hepaticae (the liverworts), and Anthocerotae (the hornworts).Since bryophytes generally lack conducting cells and a well-developed cuticle that would limit dehydration, they depend on their immediate surroundings for an adequate supply of moisture. As a result, most bryophytes live in moist or wet, shady locations, growing on rocks, trees, and soil. Some, however, have become adapted to totally aquatic habitats; others have become adapted to alternately wet and dry environments by growing during wet periods and becoming dormant during dry intervals. Although bryophytes are widely distributed, occurring in practically all parts of the world, none are found in salt water. Ecologically, some mosses are considered pioneer plants because they can invade bare areas.
Bryophytes are typically land plants but seldom attain a height of more than a few centimetres. They possess the photosynthetic pigment chlorophyll (both a and b forms) and carotenoids in cell organelles called chloroplasts. The life histories of these plants show a well-defined alternation of generations, with the independent and free-living gametophyte as the dominant photosynthetic phase in the life cycle. (This is in contrast to the vascular plants, in which the dominant photosynthetic phase is the sporophyte.) The sporophyte generation develops from, and is almost entirely parasitic on, the gametophyte. The gametophyte produces multicellular sex organs (gametangia). Female gametangia are called archegonia; (archegonium) and male gametangia, antheridia. At maturity, archegonia each contain one egg, and antheridia produce many sperm. Because the egg is retained and fertilized within the archegonium, the early stages of the developing sporophyte are protected and nourished by the gametophytic tissue. The young undifferentiated sporophyte is called an embryo. Although bryophytes have become adapted to life on land, an apparent vestige of their aquatic ancestry is that the motile (flagellated) sperm depend on water to effect gamete transport and fertilization.
Bryophytes are widely believed to have evolved from complex green algae that invaded land more than 400 million years ago. Bryophytes share some traits with green algae, such as motile sperm, similar photosynthetic pigments, and the general absence of vascular tissue. However, bryophytes have multicellular reproductive structures, whereas those of green algae are unicellular, and bryophytes are mostly terrestrial and have complex plant bodies, while the green algae are primarily aquatic and have less complex forms. An alternative view envisions the bryophytes as directly derived from early vascular plants or as an early offshoot of the lineage that gave rise to vascular plants.
Representative members
Class Musci
 moss is a term erroneously applied to many different plants (Spanish moss, a flowering plant; Irish moss, a red alga; pond moss, filamentous algae; and reindeer moss, a lichen). Mosses are classified as the class Musci in the division Bryophyta. Two major groups of mosses are recognized, the true mosses and the commercially important peat mosses (Sphagnum). (The granite mosses (granite moss) form a small group comprising the genera Andreaea and Andreaeobryum.) Sphagnum often grows in dense mats in acidic boglands. Because the peat mosses are very absorbent, they are widely used to improve soil texture and to surround plant roots during shipment and replanting in order to prevent desiccation.
moss is a term erroneously applied to many different plants (Spanish moss, a flowering plant; Irish moss, a red alga; pond moss, filamentous algae; and reindeer moss, a lichen). Mosses are classified as the class Musci in the division Bryophyta. Two major groups of mosses are recognized, the true mosses and the commercially important peat mosses (Sphagnum). (The granite mosses (granite moss) form a small group comprising the genera Andreaea and Andreaeobryum.) Sphagnum often grows in dense mats in acidic boglands. Because the peat mosses are very absorbent, they are widely used to improve soil texture and to surround plant roots during shipment and replanting in order to prevent desiccation.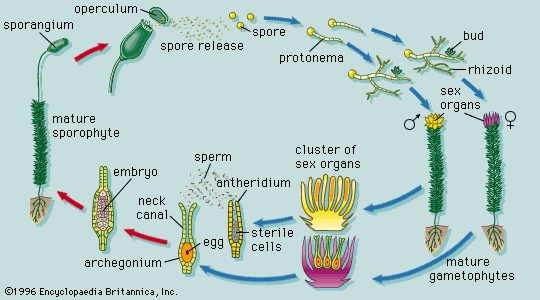 The moss gametophyte possesses leaflike structures (phyllids) that usually are a single cell layer thick, have a costa (midrib), and are spirally arranged on a stemlike axis (caulid). The moss gametophyte is an independent plant and is the familiar, erect “leafy” shoot. Multicellular rhizoids anchor the gametophyte to the substrate. The sporophyte plant develops from the tip of the fertile leafy shoot. After repeated cell divisions, the young sporophyte (embryo) transforms into a mature sporophyte consisting of foot, elongate seta, and capsule. The capsule is often covered by a calyptra, which is the enlarged remains of the archegonium. The capsule is capped by an operculum (lid), which falls off, exposing a ring of teeth (the peristome) that regulates the dispersal of spores.
The moss gametophyte possesses leaflike structures (phyllids) that usually are a single cell layer thick, have a costa (midrib), and are spirally arranged on a stemlike axis (caulid). The moss gametophyte is an independent plant and is the familiar, erect “leafy” shoot. Multicellular rhizoids anchor the gametophyte to the substrate. The sporophyte plant develops from the tip of the fertile leafy shoot. After repeated cell divisions, the young sporophyte (embryo) transforms into a mature sporophyte consisting of foot, elongate seta, and capsule. The capsule is often covered by a calyptra, which is the enlarged remains of the archegonium. The capsule is capped by an operculum (lid), which falls off, exposing a ring of teeth (the peristome) that regulates the dispersal of spores.Class Hepaticae
Liverworts (liverwort), the second major class of bryophytes, are found in the same types of habitat as mosses, and species of the two classes are often intermingled on the same site. The curious name “liverwort” is a relic of the medieval belief in the “doctrine of signatures,” which held that the external form of a plant provided a clue to which diseased body organ could be cured by a preparation made from that particular plant. There are two types of liverworts (also called hepatics) based on reproductive features and thallus structure. The more numerous “leafy” liverworts (leafy liverwort) superficially resemble mosses, but most notably differ in having lobed or divided leaves that are without a midrib and which are positioned in three rows. Thalloid (thallose) liverworts have a ribbonlike, or strap-shaped, body that grows flat on the ground. They have a high degree of internal structural differentiation into photosynthetic and storage zones. Liverwort gametophytes have unicellular rhizoids. Liverworts have an alternation of generations similar to that of mosses and, as with mosses, the gametophyte generation is dominant. The sporophytes, however, are not microscopic and are often borne on specialized structures. They sometimes resemble small umbrellas and are called antheridiophores and archegoniophores.
Class Anthocerotae
The third class of bryophytes comprises the hornworts (hornwort), a minor group numbering fewer than 100 species. The gametophyte is a small, ribbonlike thallus that resembles a thallose liverwort. The name hornwort is derived from the unique, slender, upright sporophytes, which are about three to four centimetres long at maturity and dehisce longitudinally into two valves that twist in response to changing humidity, thereby releasing spores in small numbers over a fairly long period of time.
Vascular plants
Definition of the category
 Vascular plants (tracheophytes (tracheophyte)) differ from the nonvascular bryophytes in that they possess specialized supporting and water-conducting tissue, called xylem, and food-conducting tissue, called phloem. The xylem is composed of nonliving cells (tracheids and vessel elements) that are stiffened by the presence of lignin, a hardening substance that reinforces the cellulose cell wall. The living sieve elements that comprise the phloem are not lignified. Xylem and phloem are collectively called vascular tissue and form a central column (stele) through the plant axis. The ferns, gymnosperms, and flowering plants are all vascular plants. Because they possess vascular tissues, these plants have true stems, leaves, and roots. Before the development of vascular tissues, the only plants of considerable size existed in aquatic environments where support and water conduction were not necessary. A second major difference between the vascular plants and bryophytes is that the larger, more conspicuous generation among vascular plants is the sporophytic phase of the life cycle.
Vascular plants (tracheophytes (tracheophyte)) differ from the nonvascular bryophytes in that they possess specialized supporting and water-conducting tissue, called xylem, and food-conducting tissue, called phloem. The xylem is composed of nonliving cells (tracheids and vessel elements) that are stiffened by the presence of lignin, a hardening substance that reinforces the cellulose cell wall. The living sieve elements that comprise the phloem are not lignified. Xylem and phloem are collectively called vascular tissue and form a central column (stele) through the plant axis. The ferns, gymnosperms, and flowering plants are all vascular plants. Because they possess vascular tissues, these plants have true stems, leaves, and roots. Before the development of vascular tissues, the only plants of considerable size existed in aquatic environments where support and water conduction were not necessary. A second major difference between the vascular plants and bryophytes is that the larger, more conspicuous generation among vascular plants is the sporophytic phase of the life cycle.The vegetative body of vascular plants is adapted to terrestrial life in various ways. In addition to vascular tissue, the aerial body is covered with a well-developed waxy layer ( cuticle) that decreases water loss. Gases are exchanged through numerous pores (stomata (stomate)) in the outer cell layer. The root system is involved in the uptake from the soil of water and minerals that are used by the root system as well as the stem and leaves. Roots also anchor the plant and store food. The stem conducts water and minerals absorbed by the root system upward to various parts of the stem and leaves; stems also conduct carbohydrates manufactured through the process of photosynthesis from the leaves to various parts of the stem and root system. Leaves (leaf) are supported by the stem and are oriented in a manner conducive to maximizing the amount of leaf area involved in trapping sunlight for use in photosynthesis.
 Modifications of roots, stems, and leaves have enabled species of vascular plants to survive in a variety of habitats encompassing diverse and even extreme environmental conditions. The ability of vascular plants to flourish in so many different habitats is a key factor in their having become the dominant group of terrestrial plants.
Modifications of roots, stems, and leaves have enabled species of vascular plants to survive in a variety of habitats encompassing diverse and even extreme environmental conditions. The ability of vascular plants to flourish in so many different habitats is a key factor in their having become the dominant group of terrestrial plants.The vascular plants are divisible into the nonseed plants (lower vascular plants (lower vascular plant), or cryptogams) and those that reproduce by seeds (higher vascular plants, or phanerogams). The ferns (Filicophyta) are a group of the lower vascular plants; other groups include the whisk ferns (Psilotophyta), club and spike mosses (Lycophyta), and horsetails (Sphenophyta, or Arthrophyta). Collectively, the latter four groups are sometimes referred to as pteridophytes, since each reproduces by spores liberated from dehiscent sporangia (free sporing). Although the lower vascular plants have adapted to terrestrial life, they are similar to bryophytes in that, as an apparent vestige of their aquatic ancestry, all produce motile (flagellated) male gametes (antherozoids, or sperm) and must rely on water for fertilization to be effected.
Nonseed plants
Division Filicophyta
 Ferns (fern) are a diverse group of plants technically classified in the division Filicophyta. Although they have a worldwide distribution, ferns are more common in tropical and subtropical regions. They range in size and complexity from small floating aquatic plants less than 2 centimetres long to tall tree ferns 20 metres (65 feet) high. Tropical tree ferns possess erect, columnar trunks and large, compound (divided) leaves more than 5 metres long. As a group, ferns are either terrestrial or epiphytic (growing upon another plant). Fern stems never become woody (composed of secondary tissue containing lignin) since all tissues of the plant body originate at the stem apex.
Ferns (fern) are a diverse group of plants technically classified in the division Filicophyta. Although they have a worldwide distribution, ferns are more common in tropical and subtropical regions. They range in size and complexity from small floating aquatic plants less than 2 centimetres long to tall tree ferns 20 metres (65 feet) high. Tropical tree ferns possess erect, columnar trunks and large, compound (divided) leaves more than 5 metres long. As a group, ferns are either terrestrial or epiphytic (growing upon another plant). Fern stems never become woody (composed of secondary tissue containing lignin) since all tissues of the plant body originate at the stem apex.Ferns typically possess a rhizome (horizontal stem) that grows partially underground; the deeply divided fronds (leaves) and the roots grow out of the rhizome. Fronds are characteristically coiled in the bud (fiddleheads) and uncurl in a type of leaf development called circinate vernation. Fern leaves are either whole or variously divided. The leaf types are differentiated into rachis (axis of a compound leaf), pinnae (primary divisions), and pinnules (ultimate segments of a pinna). Fern leaves often have prominent epidermal hairs and large chaffy scales. Venation of fern leaves is usually open dichotomous (forking into two equal parts).
Each frond is a potential sporophyll (spore-bearing leaf) and as such can bear structures that are associated with reproduction. When growth conditions are favourable, a series of brown patches appear on the undersurface of the sporophylls. Each one of the patches (called a sorus) is composed of many sporangia, or spore cases, which are joined by a stalk to the sporophyll. The spore case is flattened, with a layer of sterile, or nonfertile, cells surrounding the spore mother cells. Each spore mother cell divides by reduction division (meiosis) to produce haploid spores, which are shed in a way characteristic to the ferns.
Each fern spore has the potential to grow into a green, heart-shaped, independent gametophyte plant (prothallus (prothallium)) capable of photosynthesis. In contrast to bryophytes, in which the sporophyte is nutritionally dependent on the gametophyte during its entire existence, the fern sporophyte is dependent on the gametophyte for nutrition only during the early phase of its development; thereafter, the fern sporophyte is free-living. In some ferns the sexes are separate, meaning a gametophyte will bear only male or female sex organs. Other species have gametophytes bearing both sex organs. Features important in the identification of ferns include such aspects of the mature sporophyte plant as differences in the stem, frond, sporophyll, sporangium, and position of the sporangium and the absence or presence, as well as shape, of the indusium (a membranous outgrowth of the leaf) covering the sporangia.
Division Psilotophyta
 Psilotophyta (whisk ferns (whisk fern)) is a division represented by two living genera (Psilotum and Tmesipteris) and several species that are restricted to the subtropics. This unusual group of small herbaceous plants is characterized by a leafless and rootless body possessing a stem that exhibits a primitive dichotomous type of branching: it forks into equal halves. The photosynthetic function is assumed by the stem, and the underground rhizome anchors the plant. The vascular tissue is organized into a poorly developed central cylinder in the stem.
Psilotophyta (whisk ferns (whisk fern)) is a division represented by two living genera (Psilotum and Tmesipteris) and several species that are restricted to the subtropics. This unusual group of small herbaceous plants is characterized by a leafless and rootless body possessing a stem that exhibits a primitive dichotomous type of branching: it forks into equal halves. The photosynthetic function is assumed by the stem, and the underground rhizome anchors the plant. The vascular tissue is organized into a poorly developed central cylinder in the stem.Division Lycophyta (lycophyte)
This division is represented by four or more living genera, with the principal genera being Lycopodium (club mosses (club moss)), Selaginella (spike mosses (spike moss)), and Isoetes (quillworts (quillwort)). Extant members of Lycophyta occur in both temperate and tropical regions and represent the survivors of a group of vascular plants that was extremely diverse and numerous. As a group, the lycopods were prominent in the great coal-forming swamp forests of the Carboniferous Period (360 to 280 million years ago). Although all living lycopods are small herbaceous plants, some extinct types were large trees. Lycopods are differentiated into stem, root, and leaf (microphylls). Sporangia are positioned on the upper (adaxial) surface of the leaf (sporophyll). Some species form distinct cones or strobili, while others do not.
Division Sphenophyta (Equisetopsida)
 Sphenophyta (also called horsetails (horsetail) and scouring rushes) is a division represented by a single living genus (Equisetum). It has a worldwide distribution but occurs in greater variety in the Northern Hemisphere. Like the lycopods, the sphenophytes were a diverse and prominent group of vascular plants during the Carboniferous Period, when some genera attained great size in the coal-forming swamp forests. Sphenophytes are differentiated into stem, leaf (microphylls), and root. Green aerial stems have longitudinal ridges and furrows extending the length of the internodes, and stems are jointed (articulated). Surface cells are characteristically filled with silica. Branches, when they occur, are borne in whorls at the node, as are the scale leaves. Sporangia are borne in terminal strobili. The Sphenophyta are an independent line of vascular plant evolution that had its origin in the Devonian Period (408 to 360 million years ago).
Sphenophyta (also called horsetails (horsetail) and scouring rushes) is a division represented by a single living genus (Equisetum). It has a worldwide distribution but occurs in greater variety in the Northern Hemisphere. Like the lycopods, the sphenophytes were a diverse and prominent group of vascular plants during the Carboniferous Period, when some genera attained great size in the coal-forming swamp forests. Sphenophytes are differentiated into stem, leaf (microphylls), and root. Green aerial stems have longitudinal ridges and furrows extending the length of the internodes, and stems are jointed (articulated). Surface cells are characteristically filled with silica. Branches, when they occur, are borne in whorls at the node, as are the scale leaves. Sporangia are borne in terminal strobili. The Sphenophyta are an independent line of vascular plant evolution that had its origin in the Devonian Period (408 to 360 million years ago).Seed plants
 Gymnosperms and angiosperms (flowering plants) share with ferns a dominant, independent sporophyte generation; the presence of vascular tissue; differentiation of the plant body into root, stem, and leaf derived from a bipolar embryo (having stem and root-growing apexes); and similar photosynthetic pigments. Unlike ferns, however, the seed plants (spermatophyte) have stems that branch laterally and vascular tissue that is arranged in strands (bundles) around the pith (eustele). Among seed plants, as in ferns, the stem tissues that arise directly from the shoot apex are called primary tissues. Primary tissues contribute to the longitudinal growth of the stem, or primary growth. Secondary growth, resulting in an increase in the width of the axis, is produced by meristematic (meristem) tissue between the primary xylem and phloem called vascular cambium. This meristem consists of a narrow zone of cells that form new secondary xylem (wood) and secondary phloem (secondary vascular tissues).
Gymnosperms and angiosperms (flowering plants) share with ferns a dominant, independent sporophyte generation; the presence of vascular tissue; differentiation of the plant body into root, stem, and leaf derived from a bipolar embryo (having stem and root-growing apexes); and similar photosynthetic pigments. Unlike ferns, however, the seed plants (spermatophyte) have stems that branch laterally and vascular tissue that is arranged in strands (bundles) around the pith (eustele). Among seed plants, as in ferns, the stem tissues that arise directly from the shoot apex are called primary tissues. Primary tissues contribute to the longitudinal growth of the stem, or primary growth. Secondary growth, resulting in an increase in the width of the axis, is produced by meristematic (meristem) tissue between the primary xylem and phloem called vascular cambium. This meristem consists of a narrow zone of cells that form new secondary xylem (wood) and secondary phloem (secondary vascular tissues).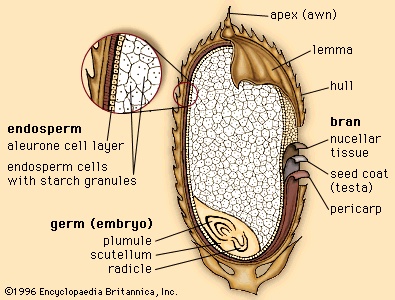 Major evolutionary advancements of these plants are demonstrated by the generally more complex plant body and by reproduction via seeds (seed and fruit). Seeds represent an important evolutionary innovation within the plant kingdom. Each seed has an embryonic plant (sporophyte), food-storage tissue, and hardened protective covering (seed coat). The seed thus contains and protects the embryonic plant and, as the primary dispersal unit of the seed plants, represents a significant improvement over the spore, with its limited capacity for survival.
Major evolutionary advancements of these plants are demonstrated by the generally more complex plant body and by reproduction via seeds (seed and fruit). Seeds represent an important evolutionary innovation within the plant kingdom. Each seed has an embryonic plant (sporophyte), food-storage tissue, and hardened protective covering (seed coat). The seed thus contains and protects the embryonic plant and, as the primary dispersal unit of the seed plants, represents a significant improvement over the spore, with its limited capacity for survival. In comparing ferns and seed plants and their life histories, certain significant differences are seen. The gametophyte in seed plants has been reduced in size, usually consisting of a few to a dozen cells. Thus, it is no longer itself a plant body, as in the bryophytes and ferns. The gametophyte is not free-living but is embedded in the sporophyte and is thus less vulnerable to environmental stress than the gametophytes of bryophytes and ferns. Finally, the spores of seed plants are male and female, as are the sporangia that contain them. The spores are not dispersed as in the bryophytes and ferns but develop into gametophytes within the sporangia. In the most advanced seed plants, the male gametes (sperm) are carried to the egg by a later extension of the pollen grain called the pollen tube. The advantage of this system is that the nonflagellated sperm are no longer dependent on water to reach the egg.
In comparing ferns and seed plants and their life histories, certain significant differences are seen. The gametophyte in seed plants has been reduced in size, usually consisting of a few to a dozen cells. Thus, it is no longer itself a plant body, as in the bryophytes and ferns. The gametophyte is not free-living but is embedded in the sporophyte and is thus less vulnerable to environmental stress than the gametophytes of bryophytes and ferns. Finally, the spores of seed plants are male and female, as are the sporangia that contain them. The spores are not dispersed as in the bryophytes and ferns but develop into gametophytes within the sporangia. In the most advanced seed plants, the male gametes (sperm) are carried to the egg by a later extension of the pollen grain called the pollen tube. The advantage of this system is that the nonflagellated sperm are no longer dependent on water to reach the egg.
 Another terrestrial adaptation of the seed plants (pollination) not found in ferns is pollen dispersed by wind or animals. Pollen is a unit of genetic material as well as part of the seed formation process. The dispersal of pollen by wind or insects, in addition to dispersal of seeds, promotes genetic recombination and distribution of the species over a wide geographic area.
Another terrestrial adaptation of the seed plants (pollination) not found in ferns is pollen dispersed by wind or animals. Pollen is a unit of genetic material as well as part of the seed formation process. The dispersal of pollen by wind or insects, in addition to dispersal of seeds, promotes genetic recombination and distribution of the species over a wide geographic area.Gymnosperms (gymnosperm)
 The term gymnosperm (“naked seeds”) represents four extant divisions of vascular plants whose ovules (seeds) are exposed on the surface of cone scales. The cone-bearing gymnosperms are among the largest and oldest living organisms in the world. They dominated the landscape about 200 million years ago. Today gymnosperms are of great economic value as major sources of lumber products, pulpwood, turpentine, and resins.
The term gymnosperm (“naked seeds”) represents four extant divisions of vascular plants whose ovules (seeds) are exposed on the surface of cone scales. The cone-bearing gymnosperms are among the largest and oldest living organisms in the world. They dominated the landscape about 200 million years ago. Today gymnosperms are of great economic value as major sources of lumber products, pulpwood, turpentine, and resins. conifer stems are composed of a woody axis containing primitive water- and mineral-conducting cells called tracheids (tracheid). Tracheids are interconnected by passages called bordered pits. Leaves are often needlelike or scalelike and typically contain canals filled with resin. The leaves of pine are borne in bundles (fascicles), and the number of leaves per fascicle is an important distinguishing feature. Most gymnosperms are evergreen, but some, such as larch and bald cypress, are deciduous (the leaves fall after one growing season). The leaves of many gymnosperms are adapted to water conservation in having a thick cuticle and in having a stomata below the leaf surface.
conifer stems are composed of a woody axis containing primitive water- and mineral-conducting cells called tracheids (tracheid). Tracheids are interconnected by passages called bordered pits. Leaves are often needlelike or scalelike and typically contain canals filled with resin. The leaves of pine are borne in bundles (fascicles), and the number of leaves per fascicle is an important distinguishing feature. Most gymnosperms are evergreen, but some, such as larch and bald cypress, are deciduous (the leaves fall after one growing season). The leaves of many gymnosperms are adapted to water conservation in having a thick cuticle and in having a stomata below the leaf surface.The tree or shrub is the sporophyte generation. In conifers, the male and female sporangia are produced on separate structures called cones or strobili. Individual trees are typically monoecious (male and female cones are borne on the same tree). A cone is a modified shoot with a single axis, on which is borne a spirally arranged series of pollen- or ovule-bearing scales or bracts. The male cone, or microstrobilus, is usually smaller than the female cone (megastrobilus) and is essentially an aggregation of many small structures (microsporophylls) that encase the pollen in microsporangia.
 The extant cycads (division Cycadophyta (cycadophyte)) are a group of ancient seed plants that are survivors of a complex which has existed since the Mesozoic Era (beginning 245 million years ago). They are presently distributed in the tropics and subtropics of both hemispheres. Cycads are palmlike in general appearance, with an unbranched, columnar trunk and a crown of large, pinnately compound (divided) leaves. The sexes are always separate, resulting in male and female plants (i.e., cycads are dioecious). Most species produce conspicuous cones (strobili) on both male and female plants, and the seeds are very large.
The extant cycads (division Cycadophyta (cycadophyte)) are a group of ancient seed plants that are survivors of a complex which has existed since the Mesozoic Era (beginning 245 million years ago). They are presently distributed in the tropics and subtropics of both hemispheres. Cycads are palmlike in general appearance, with an unbranched, columnar trunk and a crown of large, pinnately compound (divided) leaves. The sexes are always separate, resulting in male and female plants (i.e., cycads are dioecious). Most species produce conspicuous cones (strobili) on both male and female plants, and the seeds are very large. The ginkgophytes (ginkgo) (division Ginkgophyta (ginkgophyte)), although abundant, diverse, and widely distributed in the past, are represented now by a sole surviving species, Ginkgo biloba (maidenhair tree). The species was formerly restricted to southeastern China, but it is now probably extinct in the wild. The plant is commonly cultivated worldwide, however, and is particularly resistant to disease and air pollution. The ginkgo is multibranched, with stems that are differentiated into long shoots and dwarf (spur) shoots. A cluster of fan-shaped, deciduous leaves with open dichotomous venation occurs at the end of each lateral spur shoot. Sexes are separate, and distinct cones are not produced. Female trees produce plumlike seeds with a fleshy outer layer and are noted for their foul smell when mature.
The ginkgophytes (ginkgo) (division Ginkgophyta (ginkgophyte)), although abundant, diverse, and widely distributed in the past, are represented now by a sole surviving species, Ginkgo biloba (maidenhair tree). The species was formerly restricted to southeastern China, but it is now probably extinct in the wild. The plant is commonly cultivated worldwide, however, and is particularly resistant to disease and air pollution. The ginkgo is multibranched, with stems that are differentiated into long shoots and dwarf (spur) shoots. A cluster of fan-shaped, deciduous leaves with open dichotomous venation occurs at the end of each lateral spur shoot. Sexes are separate, and distinct cones are not produced. Female trees produce plumlike seeds with a fleshy outer layer and are noted for their foul smell when mature.The gnetophytes (division Gnetophyta (gnetophyte)) comprise a group of three unusual genera. Ephedra occurs as a shrub in dry regions in tropical and temperate North and South America and in Asia, from the Mediterranean Sea to China. Species of Gnetum occur as woody shrubs, vines, or broad-leaved trees and grow in moist tropical forests of South America, Africa, and Asia. Welwitschia, restricted to extreme deserts (less than 25 millimetres 【1 inch】 of rain per year) in a narrow belt about 1,000 kilometres (600 miles) long in southwestern Africa, is an unusual plant composed of an enormous underground stem and a pair of long, strap-shaped leaves that lie along the ground. The three genera differ from all other gymnosperms in possessing vessel elements (as compared to tracheids) in the xylem and in specializations in reproductive morphology. The gnetophytes have figured prominently in the theories about gymnospermous origins of the angiosperms (angiosperm).
Angiosperms


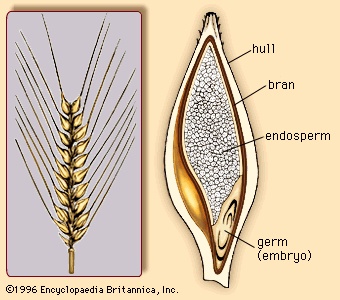 Approximately 130 million years ago flowering plants (angiosperms) evolved from gymnosperms, although the identity of the specific gymnospermous ancestral group remains unresolved. The primary distinction between gymnosperms and angiosperms is that angiosperms reproduce by means of flowers (flower). Flowers are modified shoots bearing a series of leaflike, modified appendages and containing ovules (immature seeds) surrounded and protected by the female reproductive structure, the carpel or pistil. Along with other features, angiospermy, the enclosed condition of the seed, gave the flowering plants a competitive advantage and enabled them to come to dominate the extant flora. Flowering plants have also fully exploited the use of insects and other animals as agents of pollination (the transfer of pollen from male to female floral structures). In addition, the water-conducting cells and food-conducting tissue are more complex and efficient in flowering plants than in other land plants. Finally, flowering plants possess a specialized type of nutritive tissue in the seed, endosperm. Endosperm is the chief storage tissue in the seeds of grasses; hence, it is the primary source of nutrition in corn, rice, wheat, and other cereals that have been utilized as major food sources by humans and other animals.
Approximately 130 million years ago flowering plants (angiosperms) evolved from gymnosperms, although the identity of the specific gymnospermous ancestral group remains unresolved. The primary distinction between gymnosperms and angiosperms is that angiosperms reproduce by means of flowers (flower). Flowers are modified shoots bearing a series of leaflike, modified appendages and containing ovules (immature seeds) surrounded and protected by the female reproductive structure, the carpel or pistil. Along with other features, angiospermy, the enclosed condition of the seed, gave the flowering plants a competitive advantage and enabled them to come to dominate the extant flora. Flowering plants have also fully exploited the use of insects and other animals as agents of pollination (the transfer of pollen from male to female floral structures). In addition, the water-conducting cells and food-conducting tissue are more complex and efficient in flowering plants than in other land plants. Finally, flowering plants possess a specialized type of nutritive tissue in the seed, endosperm. Endosperm is the chief storage tissue in the seeds of grasses; hence, it is the primary source of nutrition in corn, rice, wheat, and other cereals that have been utilized as major food sources by humans and other animals.Classification of angiosperms
The flowering plants are represented by two divergent evolutionary lines, the monocotyledons and the dicotyledons (dicotyledon), treated as separate classes within the division. These two major groups are distinguished by the number of embryonic seed leaves (cotyledons), arrangement of vascular tissue in the stem, leaf venation, and manner of leaf attachment to the stem. A further distinction between the two is in the number of flower parts. Generally, monocots have flower parts in multiples of three, and dicots have flower parts in multiples of four or five. The pollen of monocots is uniaperturate (with a single germinal aperture or germ pore), whereas the pollen of dicots is most commonly triaperturate or a derived form.
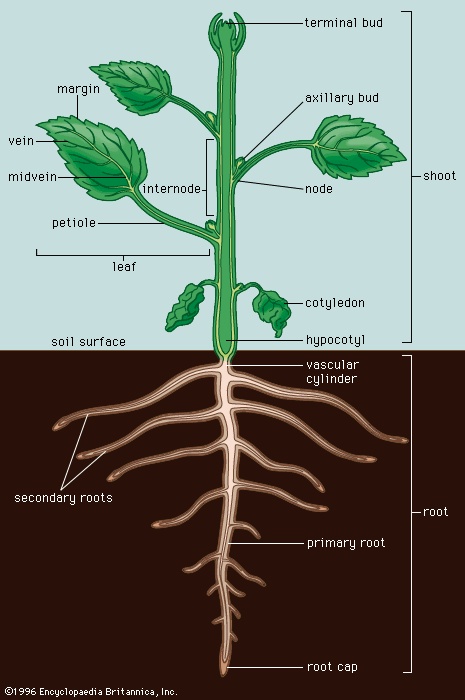 The plant body of angiosperms consists of a central axis of two parts, the shoot and root. Shoots have two kinds of organs, the stem and the leaves, while roots have one type of organ, the root itself. Systems of classification are often based upon the longevity of the portions of plant above ground. Woody plants are trees and shrubs whose shoots are durable and survive over a period of years. They are further classified into deciduous and evergreen plants. Deciduous plants drop their leaves at the end of every growing season, while evergreens keep their leaves until the leaves of the following year are produced. Herbaceous plants have soft, flexible aerial portions that die each year.
The plant body of angiosperms consists of a central axis of two parts, the shoot and root. Shoots have two kinds of organs, the stem and the leaves, while roots have one type of organ, the root itself. Systems of classification are often based upon the longevity of the portions of plant above ground. Woody plants are trees and shrubs whose shoots are durable and survive over a period of years. They are further classified into deciduous and evergreen plants. Deciduous plants drop their leaves at the end of every growing season, while evergreens keep their leaves until the leaves of the following year are produced. Herbaceous plants have soft, flexible aerial portions that die each year.Another system of classification, based on the duration of the life history, is particularly applicable to angiosperms of the temperate region. Annuals (annual) are plants that complete the entire life history (germinate from seeds, mature, flower, and produce seed) in one growing season. Examples of annuals are corn, wheat, peas, and tobacco. Biennials (biennial) complete their life history in two seasons, blooming during the second season. Beets, celery, cabbage, carrots, and turnips are biennials, but their flowers are rarely seen since they are harvested during the first season. Annuals and biennials are both generally herbaceous plants. Perennials are plants that live from year to year. Trees and shrubs are perennial, but some herbaceous plants are also perennials.
Stems (stem)


 A number of modifications of the stem occur in angiosperms, and many of these modifications provide a means for herbs to become dormant and survive for a period of years. Rhizomes (rhizome) are horizontally growing underground stems that serve as organs of asexual reproduction and food storage. Tubers (tuber) are rhizomes with thickened portions (for example, the Irish potato). Corms (corm) are short, upright underground stems surrounded by a few thin scale leaves (as in Crocus and Gladiolus). Bulbs (bulb) have a greatly reduced stem with thick, fleshy scale leaves surrounding it (as in the onion). Runners are thin, surface stems characteristic of such plants as strawberries; new plants may form on the runner as it spreads along the ground. Stolons are like runners and extend along the ground. Many of the most prolific weeds have stolons by which they propagate asexually.
A number of modifications of the stem occur in angiosperms, and many of these modifications provide a means for herbs to become dormant and survive for a period of years. Rhizomes (rhizome) are horizontally growing underground stems that serve as organs of asexual reproduction and food storage. Tubers (tuber) are rhizomes with thickened portions (for example, the Irish potato). Corms (corm) are short, upright underground stems surrounded by a few thin scale leaves (as in Crocus and Gladiolus). Bulbs (bulb) have a greatly reduced stem with thick, fleshy scale leaves surrounding it (as in the onion). Runners are thin, surface stems characteristic of such plants as strawberries; new plants may form on the runner as it spreads along the ground. Stolons are like runners and extend along the ground. Many of the most prolific weeds have stolons by which they propagate asexually.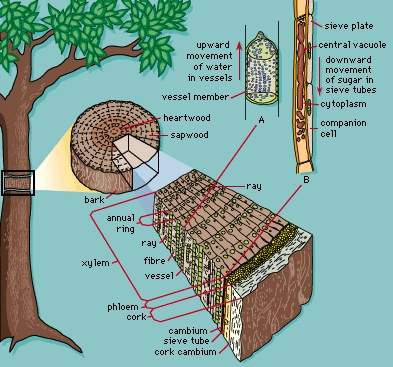 In herbaceous dicotyledonous stems the vascular conducting tissue (xylem and phloem) is organized into discrete strands or vascular bundles, each containing both xylem and phloem. The cells between the vascular bundles are thin-walled and often store starch. The peripheral region of cells in the stem is called the cortex; (cortex) cells of the central portion make up the pith. The outermost cells of the stem compose the epidermis. No bark is formed on the herbaceous stem. In contrast, woody dicot stems develop an outer layer of dead, thick-walled cells called cork cells, which together with the underlying phloem compose the bark of the tree. The major portion of the woody (wood) stem's diameter is a cylinder of xylem (wood) that originates from a region of cell division called the vascular cambium. The water-conducting cells that make up the xylem are nonliving. The accumulated xylem forms annual rings composed of two zones: a relatively wide zone of spring wood (made up of large cells, characteristic of rapid growth) and a narrower zone of summer wood (smaller cells). Xylem rays, radiating like spokes of a wagon wheel, are formed in the xylem and connect with the peripheral phloem. Stems of monocotyledons are composed of numerous vascular bundles that are arranged in a seemingly scattered manner within the ground tissue. Monocot vascular bundles lack a vascular cambium, and thus monocot stems do not become woody in a manner comparable to dicots.
In herbaceous dicotyledonous stems the vascular conducting tissue (xylem and phloem) is organized into discrete strands or vascular bundles, each containing both xylem and phloem. The cells between the vascular bundles are thin-walled and often store starch. The peripheral region of cells in the stem is called the cortex; (cortex) cells of the central portion make up the pith. The outermost cells of the stem compose the epidermis. No bark is formed on the herbaceous stem. In contrast, woody dicot stems develop an outer layer of dead, thick-walled cells called cork cells, which together with the underlying phloem compose the bark of the tree. The major portion of the woody (wood) stem's diameter is a cylinder of xylem (wood) that originates from a region of cell division called the vascular cambium. The water-conducting cells that make up the xylem are nonliving. The accumulated xylem forms annual rings composed of two zones: a relatively wide zone of spring wood (made up of large cells, characteristic of rapid growth) and a narrower zone of summer wood (smaller cells). Xylem rays, radiating like spokes of a wagon wheel, are formed in the xylem and connect with the peripheral phloem. Stems of monocotyledons are composed of numerous vascular bundles that are arranged in a seemingly scattered manner within the ground tissue. Monocot vascular bundles lack a vascular cambium, and thus monocot stems do not become woody in a manner comparable to dicots.Leaves (leaf) and roots
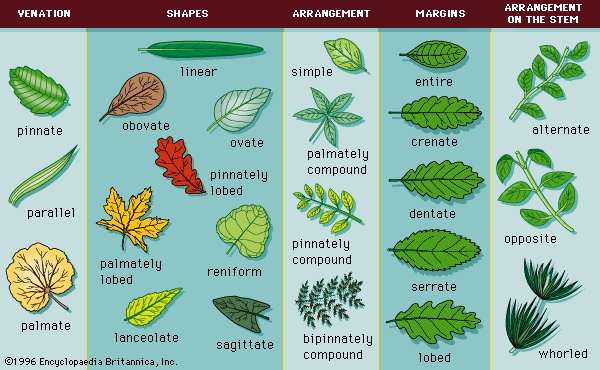 Leaves are the other plant organ that, along with stems, constitutes the shoot of the vascular plant body. Their principal function is to act as the primary site of photosynthesis in the plant. Leaves of dicots possess a network of interconnecting veins and veinlets between the larger veins of the leaf (a pattern called net venation). Leaves of monocots possess major veins that extend parallel to the long axis of the leaf (parallel venation). Leaves are classified based on leaf arrangement and on whether they are simple or compound. A leaf may be deeply lobed but still simple; a compound leaf is composed of two or more distinctly separate leaflets.
Leaves are the other plant organ that, along with stems, constitutes the shoot of the vascular plant body. Their principal function is to act as the primary site of photosynthesis in the plant. Leaves of dicots possess a network of interconnecting veins and veinlets between the larger veins of the leaf (a pattern called net venation). Leaves of monocots possess major veins that extend parallel to the long axis of the leaf (parallel venation). Leaves are classified based on leaf arrangement and on whether they are simple or compound. A leaf may be deeply lobed but still simple; a compound leaf is composed of two or more distinctly separate leaflets.Structurally, leaves are composed of an outermost layer of cells called the epidermis. Epidermal cells secrete a waxy substance (cutin) that forms a cuticle impermeable to water. The pores (stomates) in the epidermis that allow for gas exchange are formed between specialized epidermal cells called guard cells. Vascular bundles (veins) are embedded in the mesophyll, the tissue that includes all of the cells between the upper and lower epidermis. The cells of the mesophyll contain the photosynthetic pigments.
 The root system begins its development from the embryonic root (radicle), which grows out of the seed after the seed has absorbed water. This is the primary root of a new plant. The tip of the root is covered by a mass of loose cells called the root cap. Just beneath the root cap is the region of cell division of the root. Epidermal outgrowths just above the root tip are root hairs that are active in water and mineral absorption. Two types of root system are commonly distinguished, fibrous roots and taproots. Fibrous root systems are composed of large numbers of roots nearly equal in size; root systems of this type are found, for example, in the grasses. A taproot system is one in which the primary root remains the largest, and a number of smaller secondary roots are formed from it; taproots are found in such plants as carrots and dandelions. Roots that arise other than by branching from the primary roots are called adventitious roots. The prop roots of corn, for example, are adventitious.
The root system begins its development from the embryonic root (radicle), which grows out of the seed after the seed has absorbed water. This is the primary root of a new plant. The tip of the root is covered by a mass of loose cells called the root cap. Just beneath the root cap is the region of cell division of the root. Epidermal outgrowths just above the root tip are root hairs that are active in water and mineral absorption. Two types of root system are commonly distinguished, fibrous roots and taproots. Fibrous root systems are composed of large numbers of roots nearly equal in size; root systems of this type are found, for example, in the grasses. A taproot system is one in which the primary root remains the largest, and a number of smaller secondary roots are formed from it; taproots are found in such plants as carrots and dandelions. Roots that arise other than by branching from the primary roots are called adventitious roots. The prop roots of corn, for example, are adventitious.Flowers
As noted above, a primary distinction between the gymnosperms and angiosperms is that the latter have flowers. Flowers represent modified shoots that have become differentiated for reproduction. The flower bears whorls of floral organs attached to a receptacle, the expanded end of a flower stalk on which the flower parts are borne. Sepals (collectively called the calyx) are modified leaves that encase the developing flower. They are sterile floral parts and may be either green or leaflike or composed of petallike tissue. Petals (collectively called the corolla) are also sterile floral parts that usually function as visually conspicuous elements serving to attract specific pollinators to the flower. The calyx and corolla together are referred to as the perianth. Flowers that lack one or both of the above perianth parts are called incomplete. Stamens (collectively called the androecium) are the male parts of the flower. Stamens are composed of saclike anthers (microsporangia) and filaments, which are stalks that support the anthers. Anthers are usually compartmentalized and contain the pollen grains (microgametophytes). The pistil, or female part of the flower, is composed of one or a number of carpels (carpel) (collectively called the gynoecium) that fuse to form an essentially enclosed chamber. The three regions of the pistil (from the base up) are the ovary, which contains the ovules; the style, a stalked structure atop the ovary that elevates the stigma; and the stigma, a sticky knob whose surface receives the pollen during pollination.
Flowers may contain both male and female parts (a condition called perfect) or parts related to just one sex (imperfect), or they may have no sexual parts (sterile). Female and male flowers may be located on separate plants (dioecious) or on the same plant (monoecious). Flowers can also be borne singly or in aggregations called inflorescences.
Primitive flowers are radially symmetrical (actinomorphic) and characterized by numerous spirally arranged floral parts. Floral parts are free (unfused) and borne on an elongated floral axis. Sepals, petals, and stamens are attached below the ovary. Advanced flowers are bilaterally symmetrical and are characterized by a reduction in the number of floral parts. Floral parts are fused (often forming a long floral tube). Sepals, petals, and stamens are attached to the floral tube above the ovary.
Pollination is the transfer of pollen to the stigma of the same or another flower. Agents of pollination encompass a vast and diverse array of animals, including insects, birds, bats, and slugs. Flowers exhibit various adaptations to pollinators, such as showy corollas, the production of nectar (a sugary liquid), and even visual cues visible only to insects that can perceive ultraviolet wavelengths of light. Flowers pollinated by wind generally are small and lack petals. The stigma is the pollen receptor site and must be chemically compatible with any pollen that lands on it for the pollen grain to germinate. This ensures that only genetically compatible sperm are transferred to the egg.
Fruits (fruit) and seeds

 In flowering plants, ovules (ovule) are enclosed and protected in an ovary. As the ovule develops into a seed, the ovary matures into a fruit. The formation of fruits is a characteristic feature of the flowering plants. Fruits are extremely variable. In some fruits the ovary wall (pericarp) is thick and fleshy; in others it is thin and dry.
In flowering plants, ovules (ovule) are enclosed and protected in an ovary. As the ovule develops into a seed, the ovary matures into a fruit. The formation of fruits is a characteristic feature of the flowering plants. Fruits are extremely variable. In some fruits the ovary wall (pericarp) is thick and fleshy; in others it is thin and dry.
 Angiosperms have evolved many different adaptations for seed dispersal involving such agents as wind, water, and animals. Adaptations to wind dispersal include wings or plumules attached to the seed or as part of the fruit, or simply very minute seeds that are easily windborne. Adaptations to water dispersal are seeds that float or fruits that float and carry the seeds with them. Some seeds are a source of food to animals, which bury the seeds in the ground, where they later germinate. Other plants produce a fleshy fruit that is eaten along with the seeds inside it by animals, which pass the seeds through their digestive tracts unharmed. Another adaptation for animal dispersal is the development of barbed fruits or seeds that stick to the coats or skins of wandering animals. Some plants, such as witch hazels or jewelweed, can project their seeds through the air some distance from the parent plant.
Angiosperms have evolved many different adaptations for seed dispersal involving such agents as wind, water, and animals. Adaptations to wind dispersal include wings or plumules attached to the seed or as part of the fruit, or simply very minute seeds that are easily windborne. Adaptations to water dispersal are seeds that float or fruits that float and carry the seeds with them. Some seeds are a source of food to animals, which bury the seeds in the ground, where they later germinate. Other plants produce a fleshy fruit that is eaten along with the seeds inside it by animals, which pass the seeds through their digestive tracts unharmed. Another adaptation for animal dispersal is the development of barbed fruits or seeds that stick to the coats or skins of wandering animals. Some plants, such as witch hazels or jewelweed, can project their seeds through the air some distance from the parent plant.Seeds have many adaptations that enable them to survive long periods of harsh conditions. Seeds can remain viable in a dormant condition for a few days or, in some species, for hundreds of years.
reproduction and life histories
Each organism from inception to death goes through a sequence of genetically programmed developmental events constituting a life history. In eukaryotic organisms, development involves cellular events such as mitosis, meiosis, and syngamy (fertilization), which variously proceed by nuclear division (karyokinesis), cytoplasmic division (cytokinesis), cytoplasmic fusion without the union of nuclei (plasmogamy), or nuclear fusion (karyogamy).
This discussion focuses on the life histories of land plants—that is, nonvascular (bryophytes) and vascular plants, the latter comprising nonseed vascular plants (pteridophytes) and seed plants (gymnosperms and angiosperms). Although algae and fungi were traditionally regarded as plants, fungi are now universally considered as constituting the kingdom Fungi, whereas algae are often included in the kingdom Protista.
Life histories
The chromosome number in cells may be haploid, with one set of chromosomes per cell (written 1n); diploid, with two sets (2n); polyploid, with three or more sets; or dikaryotic, with a pair of nuclei in a cell (n + n), a condition that occurs mainly in fungi. Three types of sexual life histories have been recognized for the eukaryotic organisms: 1n, or haplontic; 2n, or diplontic; and 1n-2n (2n-1n). The former two types have collectively been called haplobiontic or monobiontic, because the life histories include only one phase; the third type has been called haplodiplontic, diplohaplontic, diplobiontic, dibiontic, or sporic, because the life history involves two alternating (alternation of generations) multicellular phases, or generations. Algae and fungi have many variants of all three types, especially the first, while land plants have the third type exclusively. In addition, all land plants are strictly oogamous, having motile sperm and nonmotile eggs. (In contrast, the algae and fungi may be oogamous or, frequently, isogamous or anisogamous, the latter conditions characterized by morphologically similar gametes that are either of the same size or with the female gametes of a larger size, respectively.)
The 1n-2n life history of bryophytes and vascular plants comprises the entire sequence of developmental events from zygote formation via syngamy ( fertilization) to spore formation via meiosis. Syngamy and meiosis are successive events in a sexual life history. Syngamy involves the union of two 1n gametes (gamete) to form a 2n zygote, which eventually develops into a 2n sporophyte. Meiosis involves the division of a 2n sporocyte (meiocyte, spore mother cell, pollen mother cell) to produce four 1n spores. These four spores constitute a tetrad. Gametes are 1n cells that fuse to form a zygote, whereas spores are 1n cells that develop into gameophytes without uniting with another cell.
Syngamy and meiosis are generally inseparable and alternate; consequently, the sporophyte develops from a zygote formed via syngamy, whereas the haploid gametophyte results after the sporophyte undergoes meiosis to form spores. These two phases, or generations, are multicellular in land plants. This type of life history involves an alternation of generations, a phenomenon not occurring in haplobiontic (1n or 2n) life histories. In the latter, such alternation is evident in both the morphological and the nuclear (chromosomal) changes that occur.
Meiosis and syngamy (fertilization) are the critical events that separate the sporophytic and gametophytic generations. (All nonmeiotic cell divisions involved in development are mitotic, in which chromosomes replicate, giving each daughter cell a full complement.) The zygote is the first stage and the sporocyte the last stage of the sporophytic generation, whereas the spore is the first stage and the gametes (eggs, sperm) the last stage of the gametophytic generation. The sporocytes of the multicellular 2n sporophyte divide meiotically to form 1n spores (sporogenesis). Each meiotic division results in a tetrad of four spores. (The land plants produce only sexual spores resulting from meiosis.) Each spore divides mitotically to form a multicellular 1n gametophyte, which eventually produces gametes mitotically (gametogenesis). The gametes (an egg and a sperm) fuse in the process of syngamy to form a 2n zygote. The zygote divides mitotically to form a multicellular embryo (embryogenesis), which is protected by either gametophytic tissues (such as remnants of archegonia in the nonseed land plants) or sporophytic tissues (the seed in the seed plants). An embryo, which is actually an immature sporophyte, is universally found among the land plants and often becomes dormant, but eventually grows into the mature sporophyte. During organogenesis in vascular plants the embryo develops into the mature sporophyte, with its vegetative organs (root and shoot, the latter consisting of stem and leaves) and reproductive structures (cones or strobili, flowers, etc.).
In all land plants, the two alternating generations are morphologically dissimilar, and the gametophyte is initially dominant over the sporophyte. In all bryophytes the gametophyte remains dominant, and the sporophyte is physiologically dependent on it. A homosporous life history, in which only one morphological type of spore is produced, is found in most bryophytes, although a few (e.g., Macromitrium) exhibit an anisosporous life history, in which the same sporangium produces morphologically similar spores of two different sizes.
In vascular plants, the sporophyte ultimately becomes physiologically independent of the gametophyte. A homosporous life history occurs in Psilotum (Psilotophyta), Lycopodium (Lycophyta), Equisetum (Sphenophyta), and most ferns (Filicophyta). A functionally heterosporous life history, in which the same sporangium produces morphologically similar but physiologically different spores, has been reported in a few pteridophytes—e.g., Equisetum. Finally, a heterosporous life history, in which different sporangia produce morphologically different types of spores, occurs in Selaginella, Isoetes (Lycophyta), a few aquatic ferns (Filicophyta), and all seed plants.
Homosporous life histories
A homosporous life history occurs in nearly all bryophytes and in most pteridophytes. It is characterized by morphologically identical spores that germinate to produce bisexual (both male and female) gametophytes in pteridophytes, but either bisexual or more usually unisexual (either male or female) gametophytes in bryophytes. Each mature gametophyte bears gametangia (sex organs) that produce gametes. Each antheridium (male gametangium) forms many motile, flagellate sperm, and each archegonium (female gametangium) forms one nonmotile egg. Fusion of an egg and sperm (syngamy) creates a zygote and restores the 2n ploidy level. Various mechanisms prevent the fusion of eggs and sperm from a bisexual gametophyte ( inbreeding). For example, the sex organs may mature at different times (usually antheridia mature first), or inbreeding may be chemically or genetically inhibited. The zygote divides mitotically to form the embryo, which then develops into the sporophyte. This eventually produces sporangia, which bear meiocytes (sporocytes) that divide meiotically to form spores. The number of spores produced per sporangium ranges from 16 or 32 in some pteridophytes to more than 65,000,000 in some mosses. The sporangia may be borne in specialized structures, such as sori in ferns or as cones (strobili) in many other pteridophytes. The leaflike structures that bear sporangia are called sporophylls.
In most homosporous life histories of pteridophytes the spores are both morphologically and physiologically identical and produce bisexual gametophytes. In some species of horsetail (Equisetum) the spores may be physiologically different and produce male or female gametophytes. This uncommon situation is called functional heterospory and may represent the means by which the heterosporous condition (see below) in vascular plants evolved from the homosporous condition.
Anisosporous life histories
In anisosporous life histories, an unusual phenomenon in bryophytes, there is a size difference among spores produced in the same sporangium. Each meiotic division results in a tetrad of two small spores that produce male gametophytes and two larger spores that produce female gametophytes.
Heterosporous life histories
A heterosporous life history occurs in some pteridophytes and in all seed plants. It is characterized by morphologically dissimilar spores produced from two types of sporangia: microspores, or male spores, and megaspores (macrospores), or female spores. In pteridophytes, megaspores are typically larger than microspores, but the opposite is true in most seed plants.
The spores produce two types of gametophytes: each microspore develops into a microgametophyte (male gametophyte), which ultimately produces male gametes (sperm), and each megaspore produces a megagametophyte (female gametophyte), which ultimately produces female gametes (eggs). Fusion of an egg and a sperm creates a zygote and restores the 2n ploidy level. The zygote divides mitotically to form the embryo, which then develops into the sporophyte. Eventually the sporophyte produces sporangia, which bear sporocytes (meiocytes) that undergo meiosis to form spores. Microsporangia (male sporangia) produce microsporocytes (micromeiocytes) that yield microspores. Megasporangia (female sporangia) produce megasporocytes (megameiocytes) that yield megaspores. The sporangia may be borne in specialized structures such as sori in ferns, cones (strobili) in some pteridophytes and most gymnosperms, or flowers in angiosperms. The leaflike structures bearing microsporangia and megasporangia are called, respectively, microsporophylls and megasporophylls. In angiosperms these sporophylls represent, respectively, the stamens and carpels of the flower; in gymnosperms these sporophylls may constitute parts of, respectively, microstrobili (male cones, or pollen cones) and megastrobili (female cones, ovule cones, or seed cones).
The essential difference between the homosporous and heterosporous life history is the presence in the latter of two spore types (microspores and megaspores) and their concomitant precursory structures (microsporocytes and megasporocytes; microsporangia and megasporangia; etc.) and subsequent structures (microgametophytes and megagametophytes).
Variations involving seed plants
The gymnosperms and angiosperms not only lack some reproductive structures found in the homosporous and heterosporous pteridophytes but also have certain reproductive structures peculiar to the seed plants. Heterosporous pteridophytes, like their homosporous counterparts, have archegonia, antheridia, and motile, flagellate sperm. The seed plants completely lack antheridia, and of the extant groups only the ginkgo and the cycads have flagellate sperm. Archegonia occur in most gymnosperms except Gnetum and Welwitschia, but they are lacking in all angiosperms.
Pollen grains and pollen tubes (male reproductive structures), ovules and seeds (female reproductive structures), and seedlings are structures unique to all seed plants. The ovule is a single megasporangium (in seed plants this is called the nucellus) surrounded by one or two integuments (in rare cases, none or three) and containing inside the nucellus a single megasporocyte (spore mother cell). The megasporocyte undergoes meiosis to form four megaspores, three of which typically degenerate, the remaining one developing into the megagametophyte (female gametophyte). Ovules never dehisce to release their megaspores, unlike the megasporangia of most pteridophytes. The pollen grain is the partly or completely developed microgametophyte (male gametophyte). It is usually multicellular, consisting of two or three cells in angiosperms and usually two to five cells in gymnosperms, although in conifers it is occasionally one cell (for example, the families Taxaceae and some Cupressaceae) or 6 to 43 cells (the families Araucariaceae and some Podocarpaceae).
During pollination, pollen is transferred from its source to a receptive surface: in gymnosperms from the microsporangium to the integument or, especially, the pollination droplet of the ovule (rarely to the cone scale); in angiosperms from the microsporangium (pollen sac) of the anther to the stigma of the carpel. Once pollen has reached the appropriate receptive source, it germinates to form the pollen tube, a structure that grows toward the megagametophyte and in so doing conveys the sperm directly to the egg. All angiosperms and most gymnosperms, except the ginkgo, cycads, and some fossil seed plants, lack swimming sperm. The presence of swimming sperm apparently represents a more primitive, transitional evolutionary condition. After fertilization, the ovule transforms into a seed. The integument or integuments become modified into the seed coat. The seed (seed and fruit) typically becomes dormant for a period of time before it germinates to produce a seedling.
Double fertilization is a phenomenon unique to angiosperms. Each pollen grain produces two sperm; one fuses with an egg to form the zygote, and the other fuses with one or more polar nuclei in the female gametophyte (megagametophyte, or also “embryo sac”) to form an endosperm, which has a ploidy level that varies from 2n to 15n. In approximately 70 percent of the known cases, the second sperm fuses with two endosperm nuclei to produce a 3n (triploid) endosperm. The endosperm is a special nutritive tissue for the embryo and, after seed germination, for the seedling. In contrast, the megagametophyte is the comparable nutritive tissue in the gymnosperms.
Asexual reproduction
Both homosporous and heterosporous life histories may exhibit various types of asexual reproduction (vegetative reproduction, somatic reproduction). Asexual reproduction is any reproductive process that does not involve meiosis or the union of nuclei, sex cells, or sex organs. Depending on the type of life history, asexual reproduction can involve the 1n or 2n generation.
The significance of sexual reproduction is that it is responsible for the genetic variation arising in a population as a result of the segregation and recombination of genetic material via meiosis and syngamy, respectively (the cells that result from sexual reproduction are genetically different from their parent cells). The significance of asexual reproduction is that it is a means for a rapid and significant increase in the numbers of individuals. (Weeds, for instance, are successful partly due to their great capacity for vegetative reproduction.) The cells that result from asexual reproduction are genetically identical to their parent cells. In addition, vegetative reproduction in the bryophytes and pteridophytes is a means of bypassing the somewhat lengthy and moisture-dependent sexual process—that is, the motile, swimming sperm characteristic of these groups require the presence of water, which may be a limiting factor in drier times.
Deviations from the usual life history
In most life histories, a 2n sporophyte typically alternates with a 1n gametophyte, but there are significant deviations. Apospory is the development of 2n gametophytes, without meiosis and spores, from vegetative, or nonreproductive, cells of the sporophyte. In contrast, apogamy is the development of 1n sporophytes without gametes and syngamy from vegetative cells of the gametophyte. The 2n aposporous gametophytes and 1n apogamous sporophytes are usually infertile under natural conditions because of disruption of cytological events. Various compensating genetic mechanisms, however, may occur to complete the life history. parthenogenesis is the formation of a 1n embryo directly from an unfertilized egg. Apospory and apogamy occur in bryophytes, pteridophytes, and angiosperms, whereas parthenogenesis occurs in ferns and angiosperms. Apogamy is more common in pteridophytes, but apospory is more common in bryophytes.
Some ferns (certain species of Trichomanes and Vittaria) have lost the ability to produce sporophytes. The species exist as gametophytes that spread by gemmae (units of asexual reproduction); although gametangia are produced, no sporophytes result.
Plant physiology
General features of plant nutrition
Plant nutrition includes the nutrients necessary for the growth, maintenance, and reproduction of individual plants; the mechanisms by which plants acquire such nutrients; and the structural, physiological, and biochemical roles these nutrients play in metabolism.
Mode of nutrition
All organisms obtain their nutrients from the environment, but not all organisms require the same nutrients, nor do they assimilate these nutrients in the same way. There are two basic nutritional types, autotrophs and heterotrophs. Heterotrophs require both inorganic and organic (carbon-containing) compounds as nutrient sources. Autotrophs obtain their nutrients from inorganic compounds, and their sole source of carbon is carbon dioxide (CO2). An autotroph is photoautotrophic if light energy is required to assimilate CO2 into the organic constituents of the cell. Furthermore, a photoautotroph that also uses water as a nutrient source and liberates oxygen in the energy-trapping process of photosynthesis is an oxygenic photoautotroph. The Earth's first such organisms are believed to have been the major sources of the present-day oxygen content of the atmosphere (approximately 21 percent). All plants, as well as many monerans and protists, are characteristically oxygenic photoautotrophs.
Plants, as autotrophic organisms, use light energy to photosynthesize sugars from CO2 and water. They also synthesize amino acids and vitamins from carbon fixed in photosynthesis and from inorganic elements garnered from the environment. (Animals, as heterotrophic organisms, cannot synthesize many nutrients, including certain amino acids and vitamins, and so must take them from the environment.)
Essential elements and minerals
Certain key elements are required, or essential, for the complex processes of metabolism to take place in plants. Plant physiologists generally consider an element to be essential if the plant is unable to complete its life cycle (i.e., grow and reproduce) in its absence; the particular structural, physiological, or biochemical roles of the element cannot be satisfied by any other element; and the element is directly involved in the plant's metabolism (e.g., as part of an enzyme or other essential organic cellular constituent). Beneficial elements are those that stimulate plant growth by ameliorating the toxic effects of other elements or by substituting for an element in a less essential role (e.g., as a nonspecific osmotic solute). Some elements (chemical element) are beneficial in that they are necessary for the growth of some, but not all, plant species.
The required concentrations of each essential and beneficial element vary over a wide range. The essential elements required in relatively large quantities for adequate growth are called macroelements. Nine minerals make up this group: carbon (C), hydrogen (H), oxygen (O), nitrogen (N), potassium (K), calcium (Ca), magnesium (Mg), phosphorus (P), and sulfur (S). Seven other essential mineral elements are required in smaller amounts (0.01 percent or less) and are called microelements. These are iron (Fe), chlorine (Cl), manganese (Mn), boron (B), copper (Cu), molybdenum (Mo), and zinc (Zn). The specific required percentages may vary considerably with species, genotype (or variety), age of the plant, and environmental conditions of growth.
A macronutrient is the actual chemical form or compound in which the macroelement enters the root system of a plant. The macronutrient source of the macroelement nitrogen, for example, is the nitrate ion (NO3−). carbon dioxide from the atmosphere provides the carbon atoms and two-thirds of the oxygen required by plants. water taken from the soil provides about one-third of the oxygen and much of the hydrogen. Soil provides macroelements and microelements from mineral complexes, parent rock, and decaying organisms. Factors that determine plant root uptake include the solubility of the chemical in question, the adsorptive properties of the charged soil surfaces, and the metabolic uptake capabilities of the individual plant.
The macroelements carbon, hydrogen, oxygen, and nitrogen constitute more than 96 percent of the dry weight of plants. Thus, they are the major constituents of the structural and metabolic compounds of the plant. Their presence within cells also helps regulate osmotic pressure. Microelements are generally either activators or components of enzymes, although the macroelement potassium also serves these roles.
General overview of metabolic cycles
Metabolism denotes the sum of the chemical reactions in the cell that provide the energy and synthesized materials required for growth, reproduction, and maintenance of structure and function. In plants the ultimate source of all organic chemicals and the energy stored in their chemical bonds is the conversion of CO2 into organic compounds (CO2 fixation) by either photosynthesis or chemosynthesis. The general and specific features of plant metabolism ultimately derive from oxygenic photosynthesis, which underlies the autotrophic nutrition of plants.
Pathways and cycles
Chemical reactions in the cell occur in a sequence of stages called a metabolic pathway. Each stage is catalyzed by an enzyme, a protein that changes (usually increases) the rate at which the reaction proceeds but does not alter the reactants or end products. Certain thermodynamic conditions must be met for a reaction to proceed, even in the presence of enzymes. If the end product of the reaction is also the reactant (or substrate) that starts the pathway, then the sequence of reactions is called a metabolic cycle. The intermediate chemicals that are formed and used in the various stages of the sequence are called intermediary metabolites.
Metabolic pathways and cycles are either catabolic (energy-releasing) or anabolic (energy-consuming). Catabolic (catabolism) reactions break down complex metabolites into simpler ones, while anabolic reactions (anabolism) build up (biosynthesize) new molecules. When chemical bonds are broken, energy is released, which drives anabolic reactions to form new bonds. The energy released generally has been stored in high-energy bonds of an intermediate energy carrier molecule, such as the terminal phosphate bond of adenosine triphosphate (ATP). (When the terminal phosphate is split from the ATP molecule, adenosine diphosphate, or ADP, is formed and inorganic phosphate is released, along with energy.) The simpler metabolites formed via catabolic reactions are often the building-block metabolites used in anabolic reactions to synthesize more complex molecules (e.g., starch, proteins, or lipids).
Control mechanisms
The cells of all plants are eukaryotic, since they possess a nucleus and membrane-bound organelles (organelle), such as chloroplasts, mitochondria, glyoxyosomes, peroxisomes, and vacuoles. The thousands of metabolic reactions that take place in the cell are regulated within these organelles and their subcompartments. When compared with cells of other eukaryotic organisms, plant cells may have the highest degree of metabolic compartmentalization.
The primary mechanism of metabolic control, however, remains the enzymes themselves. Although all enzymes of the pathway help determine the net and directional flow of carbon, certain key stages are controlled by regulatory enzymes. Regulatory enzymes may catalyze either the first stage in the metabolic pathway, or they may catalyze reactions in which key branch points occur. The activity of such enzymes, in turn, may be controlled by the amount synthesized (coarse control)—in that further action by the enzyme is inhibited when some critical concentration of the reaction product is reached—or by special metabolites, called effectors, that interact directly with the enzyme (fine control). The latter metabolites may be either part of, or totally unrelated to, the metabolites of the pathway.
Another mechanism by which metabolic reactions are regulated is through transport systems in the membranes of organelles. These systems control the nature, direction, and amount of metabolites entering the metabolic pathways and are often uniquely related to the autotrophic nutrition of plants.
Principal pathways and cycles
The 6-carbon sugar glucose, a product of photosynthesis, may be translocated in the form of sucrose (a 12-carbon sugar) to nourish nonphotosynthesizing parts of the plant, or it may be polymerized into starch for storage. (Trehalose, another 12-carbon sugar, replaces sucrose in some nonvascular plants.) When required, sucrose and starch are hydrolyzed to glucose and then enter glycolysis or the pentose phosphate pathway. The reactions of both pathways take place in the cytoplasm of the cell.
The net result of glycolysis is the metabolism of glucose into two molecules of the three-carbon organic acid pyruvate. This 10-reaction metabolic pathway involves phosphate-containing intermediates and is regulated by two enzymes, which catalyze those reactions that contain the substrates fructose phosphate and phosphoenolpyruvate (PEP). Glycolysis yields ATP molecules and hydrogen; the latter is accepted by the coenzyme (coenzymes are smaller, nonprotein participants associated with certain enzymes) nicotinamide adenine dinucleotide (NAD) to form NADH. The hydrogen on NADH then reacts either with molecular oxygen (O2) to catalyze the release of energy (trapped in the high-energy bonds of ATP) or with another metabolite to reduce the molecule by the addition of hydrogen. Some intermediates are used in the biosyntheses of fat or certain amino acids.
The pentose phosphate pathway, an alternative pathway for the catabolism of glucose, operates in the presence of oxygen. It furnishes reducing power (i.e., it accepts hydrogen atoms and carries them on the coenzyme nicotinamide adenine dinucleotide phosphate 【NADP】) for use in the synthesis of fat and five-carbon (pentose) sugars that are required as components of nucleic acids, some vitamins, and key metabolites. It is regulated by the rate at which the product of the pentose pathway, NADPH, is oxidized. The end products are cycled back into the glycolytic pathway.
Pyruvate is transported into the mitochondria (mitochondrion), where it enters a sequence of 10 reactions called the tricarboxylic acid (TCA) cycle, or Krebs cycle (tricarboxylic acid cycle). Pyruvate is metabolized into the two-carbon intermediate, acetyl coenzyme A (CoA), which combines with a four-carbon acid, oxaloacetate. The product, citrate, has three acid (or carboxylic) groups; hence the name tricarboxylic acid cycle. Citrate is systematically catabolized (broken down) with progressive losses of successive carbon atoms as CO2 into five-carbon and, finally, four-carbon, acids. The latter acid, oxaloacetate, begins the cycle again. With each oxidation reaction, a hydrogen atom is transferred to the coenzyme NAD or, in one reaction, the coenzyme flavin adenine dinucleotide (FAD) to form NADH and FADH, respectively. The reduced coenzymes NADH and FADH enter into a sequence of reactions called the respiratory chain on the inner membrane of the mitochondrion. This chain is a series of carriers (ubiquinone and several iron-containing chemicals called cytochromes) that ultimately transfer the hydrogen and electrons of these coenzymes to molecular oxygen, forming water. The energy generated from the oxidation by the respiratory chain is trapped in the three ATP molecules formed per NADH molecule oxidized. The mechanism is chemiosmotic in that it involves building a hydrogen ion (proton) gradient on one side of the mitochondrial membrane.
A net of 36 ATP molecules are gained from all hydrogen-carrying coenzymes formed in glycolysis and the TCA cycle, and they represent the principal energy source for most anabolic (biosynthetic) reactions in plants. In addition, the TCA cycle furnishes metabolites for the biosynthesis of important organic molecules of the cell.
Another metabolic cycle, the isoprenoid pathway, produces essential oils, carotenoid pigments, certain plant hormones, and rubber. These metabolites (often called secondary metabolites) are unique to plants and serve such functions as attracting pollinating insects, photosynthesis, and growth and development. Plant seedlings use the glyoxylic acid cycle to convert fats (principally from seeds) into glucose. This occurs initially in the glyoxysome and subsequently in the mitochondria and cytoplasm.
Unique features of plant metabolism
The pathways outlined above exist in essentially the same form in all organisms, but metabolism in plants does have certain unique features. Plant mitochondria, for example, have specific transport systems for the NADH produced in glycolysis and for the oxaloacetate produced from a direct fixation of CO2 into PEP. Unlike animal mitochondria, plant mitochondria metabolize malate and the amino acid glycine. A special enzyme converts malate to pyruvate, thereby allowing an alternative to the glycolytic pathway. Glycine is a product of the unique plant pathway of photorespiration II (see below Photosynthesis).
Plant mitochondria possess a cyanide-resistant cytochrome-respiratory chain in addition to the cyanide-sensitive respiratory chain also found in other organisms. Oxidation of NADH through this alternative pathway produces energy in the form of heat, but no ATP. Some physiologists suggest that this pathway is a mechanism for burning off excess carbohydrate. Others believe that this pathway allows a TCA cycle, in times of increased need, to produce more than the usual amount of metabolites for biosyntheses, which, in the presence of ATP acting as a feedback inhibitor, could not normally be produced. This system functions at a high rate in the flowers of the arum (Araceae) lily. Temperatures of this organ may reach 40° C (104° F) and may contribute to the attraction of pollinators. The overall function of the cyanide-insensitive respiratory pathway, however, is not clearly understood.
photosynthesis
As discussed above, the autotrophic mode of nutrition of plants is derived from oxygenic photosynthesis. Energy-rich organic compounds are synthesized from low-energy atmospheric CO2 using the energy of absorbed sunlight. (Some bacteria are nonoxygenic photosynthesizers, utilizing hydrogen sulfide, H2S, rather than water.) The resultant organic compounds initiate the flow of energy and carbon through the food chains of agricultural and natural ecosystems, intrinsically linking plants with the heterotrophic life-forms of the remaining four kingdoms of organisms. The oxygen liberated by plants (and certain photosynthetic protists and monerans) over geologic time, has oxygenated the Earth's atmosphere and has produced fossil fuels such as coal, gas, and oil.
This section describes the basic mechanisms of photosynthesis—the acquisition of energy and the fixation of carbon dioxide—used by plants of diverse evolutionary lines.
Basic mechanisms
Electromagnetic radiation having wavelengths between approximately 430 and 700 nanometres can be seen as light by the eye and constitutes the range absorbed by plants for photosynthesis. Blue light has a wavelength around 450 nanometres, and red light, a wavelength of 650–700 nanometres.
Double-membraned cell organelles called chloroplasts (chloroplast) contain the photosynthetic apparatus: light-absorbing pigments, other electron-carrying chemicals (cytochromes and quinones), and enzymes. (Pigments absorb light of a particular wavelength; those wavelengths that are not absorbed are reflected and may be perceived as colour; hence, for example, the green colour of many plants.) The inner membrane of the chloroplast is folded into flat tubes, the edges of which are joined to hollow, sacklike disks called thylakoids. Stacks of thylakoids embedded with pigment molecules are called grana. The inner matrix of the chloroplast is called the stroma.
Photosynthesis consists of two interdependent series of reactions, the photochemical light reactions and the metabolic dark reactions; the former are dependent on light, the latter on temperature. Light reactions occur in the grana and dark reactions in the stroma. The overall formula for photosynthesis is:
6CO2 + 12H2O → C6H12O6 + 6O2 + 6H2O.
(glucose)
The light reactions (photochemical reaction), the first stage of photosynthesis, convert light energy into chemical energy (ATP and NADPH). Light reactions comprise two interdependent systems, called photosystems I and II. The dark reactions, the second stage of photosynthesis, use the chemical energy products of the light reactions to convert carbon from carbon dioxide to simple sugars.
Light reactions consist of several hundred light-absorbing pigment molecules so arranged as to maximize the gathering of light energy. These “antennae” are coupled to a minicircuit of electron-carrying chemicals. The pigments are chlorophyll a and chlorophyll b and various carotenoids. Absorbed light energy is transferred to specialized chlorophyll molecules called P700 and P680 in photosystems I and II, respectively. Once these specialized chlorophyll molecules acquire sufficient energy, electrons are given up to the electron carriers within their photosystems, initiating an electron flow. (The carrier molecules include quinones and cytochromes.) The effect of this, when photosystems I and II function synchronously, is the formation of a chemiosmotic gradient of protons that phosphorylates (adds a phosphate group to) ADP, resulting in ATP. These electrons also effect the formation of NADPH from NADP. The P680 chlorophyll, upon loss of its electron, becomes a strong oxidizing agent that subsequently causes the water molecule to dissociate into protons and oxygen gas.
The dark reactions are responsible for the conversion of carbon dioxide to glucose. The essential reaction involves the combining of CO2 with the five-carbon sugar ribulose 1,5-bisphosphate (RuBP) in a series of reactions called the Calvin-Benson cycle. This reaction yields an unstable intermediate, which breaks down into two molecules of phosphoglycerate (PGA), a three-carbon acid. Each reaction is catalyzed by a specific enzyme. Six revolutions of the cycle means that six CO2 molecules react with six RuBP molecules to produce 12 molecules of PGA; two three-carbon PGA molecules combine to form the six-carbon glucose, and 10 PGAs are recycled to regenerate six molecules of RuBP. The ATP and NADPH from light reactions provide the energy and reducing power to form glucose and refurbish the CO2 acceptor, RUBP. For further information about Calvin's work, see photosynthesis.
Specific variations in photosynthesis
Chlorophylls a and b (bound to a protein) and carotenes constitute the principal light-absorbing complex of most plants. Differences in chloroplast structure, though not major, occur among phylogenetically diverse plant groups. All such variations, however, represent evolutionary adaptations to utilize more efficiently the light energy that drives the reactions common to all oxygenic photosynthesizers, i.e., photosystems I and II.
The enzyme ribulose 1,5-bisphosphate carboxylase/oxygenase (rubisco) catalyzes the formation of organic molecules from CO2. As the major enzyme of all photosynthetic cells, rubisco is probably the most abundant protein on the Earth. There is, however, a major catalytic flaw in the ability of this enzyme to convert CO2 to sugars. In the presence of molecular oxygen, rubisco catalyzes reactions in which oxygen is introduced (i.e., it acts as an oxygenase), and CO2 is formed rather than converted.
Rubisco evolved in photosynthetic organisms that lived in the atmosphere of primitive Earth, an atmosphere which contained only traces of molecular oxygen. As photosynthesis in the Cyanobacteria of Precambrian times (Precambrian time) (3.8 billion to 570 million years ago) oxygenated the atmosphere, the ratio of carbon dioxide to oxygen fell drastically, and rubisco began to function more and more as an oxygenase. This greatly reduces the net fixation of CO2 into sugars and, therefore, photosynthetic efficiency. Rubisco as an oxygenase only splits RuBP into PGA and the two-carbon acid phosphoglycolate, which initiates the photorespiratory carbon-oxidation cycle, or photorespiration. (This cycle probably evolved to recycle PGA back into the photosynthetic pathway, thereby preventing an even greater loss of carbon.) Photorespiration involves three organelles (chloroplasts, peroxisomes, and mitochondria), each with unique transport mechanisms for the cycle's intermediates.
All plants are classified as C3 (plants that use only the Calvin-Benson cycle), C4 (plants that use an additional CO2-fixation mechanism and the Calvin-Benson cycle), C3-C4 (plants intermediate between C3 and C4), and CAM (plants that have a variant of the C4 pathway).
The majority of plants fix CO2 directly into RuBP, and their first stable product is the three-carbon acid PGA, hence the designation C3. These plants have an active photorespiratory cycle, especially in high light intensity and warm temperatures.
Sometime during the later periods of the Cenozoic Era (66.4 million years ago to the present), certain of the angiosperms (grasses and the dicotyledonous plants) of mainly tropical climates evolved a -fixation system that precedes the Calvin-Benson cycle. The first fixation is into the three-carbon acid phosphoenolpyruvate (PEP) by PEP carboxylase (an enzyme that has no oxygenase function) in the outer mesophyll cells of the leaf. The first stable fixation product is the four-carbon acid oxaloacetate, hence the designation C4 plants. Oxaloacetate is converted to malate, which is transferred to a thick-walled bundle sheath cell that shields the subsequent reactions from the high concentration of molecular oxygen in the atmosphere. Malate is decarboxylated, and rubisco of the primitive and susceptible Calvin-Benson cycle functions more efficiently because here photorespiration is suppressed. There is thus a spatial separation of initial CO2 fixation and the Calvin-Benson cycle. This efficiency is not without cost, however, as an additional ATP is required to recycle PEP. For this reason, C3 plants may be more efficient in cold climates with less light (and, therefore, less photorespiration).
There are also plants with enzymatic and leaf anatomical characteristics intermediate between C3 and C4 plants, called C3-C4 intermediate species. These plants are thought to be in the pathway of evolution to full C4 photosynthetic status.
succulent plants of the desert regions (e.g., cacti) also initially fix CO2 into oxaloacetate. This occurs only at night when conditions are cooler, however. Normally, the stomates (stomate) in leaves or stems, through which plants lose water and acquire carbon dioxide, are open in the day and closed at night; those of the succulent plants do the opposite through a special mechanism that prevents great loss of water during the hot days. The resultant oxaloacetate is converted into malate, stored in the vacuole, and released during the day when the stomates are closed. Malate is decarboxylated, and the CO2 that released is fixed by rubisco in the usual Calvin-Benson cycle. Both the C4 and C3 processes take place in the same cell. This process is called crassulacean acid metabolism (hence CAM plants) after a family of succulent plants (Crassulaceae).
Ecology
Plant geography
 Plants occur over the surface of the Earth in well-defined patterns that are closely correlated with both climate and the history of the planet. Forests (forest) are the most important of these natural communities from the standpoint of area, carbon content, annual carbon fixation, the cycling of nutrient elements, and influence on energy and water budgets, as well as being the principal reservoir of biotic diversity on land. The most extensive forests are the boreal coniferous forests of North America, Scandinavia, northern Europe, and northern Asia. The moist forests of the tropics are the most diverse, often containing as many as 100 species of trees per hectare (1 hectare = 2.47 acres) and occasionally many more.
Plants occur over the surface of the Earth in well-defined patterns that are closely correlated with both climate and the history of the planet. Forests (forest) are the most important of these natural communities from the standpoint of area, carbon content, annual carbon fixation, the cycling of nutrient elements, and influence on energy and water budgets, as well as being the principal reservoir of biotic diversity on land. The most extensive forests are the boreal coniferous forests of North America, Scandinavia, northern Europe, and northern Asia. The moist forests of the tropics are the most diverse, often containing as many as 100 species of trees per hectare (1 hectare = 2.47 acres) and occasionally many more.
 Forests are commonly distinguished from woodlands and savannas in having a closed canopy of trees that may be 10 metres to more than 50 metres in height. At their wetter and cooler limits, forests are replaced by tundra, a community of low-growing trees and shrubs with a ground cover of Sphagnum and other mosses, dwarfed shrubs, and perennial herbs. The transition from forest to tundra occurs at high latitudes and elevations and may extend over a considerable distance, throughout which trees are scattered, restricted to sheltered areas, and stunted in growth. Such taiga (boreal forest) stands are common at the northern limit of trees throughout the Northern Hemisphere. The alpine transition tends to be more abrupt. Both are often determined locally by the distribution of snow.
Forests are commonly distinguished from woodlands and savannas in having a closed canopy of trees that may be 10 metres to more than 50 metres in height. At their wetter and cooler limits, forests are replaced by tundra, a community of low-growing trees and shrubs with a ground cover of Sphagnum and other mosses, dwarfed shrubs, and perennial herbs. The transition from forest to tundra occurs at high latitudes and elevations and may extend over a considerable distance, throughout which trees are scattered, restricted to sheltered areas, and stunted in growth. Such taiga (boreal forest) stands are common at the northern limit of trees throughout the Northern Hemisphere. The alpine transition tends to be more abrupt. Both are often determined locally by the distribution of snow. Forests yield at their warmer and drier margins to grassland (called prairie in North America and steppe in Asia). In North America the rich grasslands of the eastern mid-continent, which is affected by moisture moving northward from the Gulf of Mexico, once supported a diversity of herbaceous plants, including grasses that might reach several feet in height, and were known as tall-grass prairie. Westward, where water was less abundant, the grasses diminished in diversity and size to form the short-grass plains. Southward and westward the plains yielded under increasing aridity to desert, as old as any other zone in an evolutionary sense and very diverse in species. Similar patterns of vegetation occur across the broad waist of Asia.
Forests yield at their warmer and drier margins to grassland (called prairie in North America and steppe in Asia). In North America the rich grasslands of the eastern mid-continent, which is affected by moisture moving northward from the Gulf of Mexico, once supported a diversity of herbaceous plants, including grasses that might reach several feet in height, and were known as tall-grass prairie. Westward, where water was less abundant, the grasses diminished in diversity and size to form the short-grass plains. Southward and westward the plains yielded under increasing aridity to desert, as old as any other zone in an evolutionary sense and very diverse in species. Similar patterns of vegetation occur across the broad waist of Asia. The distinction between grassland and forest is blurred in many places, especially in the semiarid subtropics and in other regions where fires may be common. This transitional zone is savanna, an open, grassed woodland. The southeastern coastal plain of North America was originally such a region, a pine savanna that produced plants adapted to, in fact dependent on, burning for survival. The long-leaf pine (Pinus palustris), for instance, has a “grass” stage, which lasts for several years of early growth, with the bud protected at the very surface of the ground by a thick tuft of long, grasslike leaves that shield it from the heat of a fire. Once sufficient energy has been stored in the taproot and short stem, the tree virtually explodes into growth and passes rapidly through a period of vulnerability to fire. As a tree of 3 metres (10 feet) or more in height, it is safe from all but the hottest of fires. Oaks and other species are replacing pines as savannas disappear from the southeastern United States because of the continuing division of land ownership and protection from fire.
The distinction between grassland and forest is blurred in many places, especially in the semiarid subtropics and in other regions where fires may be common. This transitional zone is savanna, an open, grassed woodland. The southeastern coastal plain of North America was originally such a region, a pine savanna that produced plants adapted to, in fact dependent on, burning for survival. The long-leaf pine (Pinus palustris), for instance, has a “grass” stage, which lasts for several years of early growth, with the bud protected at the very surface of the ground by a thick tuft of long, grasslike leaves that shield it from the heat of a fire. Once sufficient energy has been stored in the taproot and short stem, the tree virtually explodes into growth and passes rapidly through a period of vulnerability to fire. As a tree of 3 metres (10 feet) or more in height, it is safe from all but the hottest of fires. Oaks and other species are replacing pines as savannas disappear from the southeastern United States because of the continuing division of land ownership and protection from fire.In contrast, the extensive savannas of eastern Africa have been maintained through climate, fires, and grazing and support an extraordinary diversity of migratory animals that have evolved to use different parts of the ecosystem. The patterns of use are complex and phased in time, and they work to maintain both the diversity and productivity of the savanna. These African populations are the last of the large and diverse mammalian and avian populations that once also grazed similar areas of North America, Europe, and Asia.
 The forests (tropical rainforest) of the moist tropics remain the most complex and fascinating of terrestrial ecosystems. They extend from mountain slopes to river swamps and occurred throughout the tropics wherever precipitation allowed trees to survive. Human activities have virtually eliminated forests from vast areas of the tropics and threaten to destroy the residual forests globally. The largest areas of tropical forest occur now in the Amazon basin of South America and in the Congo basin of west-central Africa. The extensive tropical forests of Southeast Asia have been reduced to fragments of the area they occupied as recently as 1950.
The forests (tropical rainforest) of the moist tropics remain the most complex and fascinating of terrestrial ecosystems. They extend from mountain slopes to river swamps and occurred throughout the tropics wherever precipitation allowed trees to survive. Human activities have virtually eliminated forests from vast areas of the tropics and threaten to destroy the residual forests globally. The largest areas of tropical forest occur now in the Amazon basin of South America and in the Congo basin of west-central Africa. The extensive tropical forests of Southeast Asia have been reduced to fragments of the area they occupied as recently as 1950.The forests of the Amazon basin have evolved as a part of a river system whose water level fluctuates annually by as much as 15 metres (50 feet) or more along the middle and lower Amazon. There are substantial further differences in the quality of water. The Negro River, for example, drains an area of sands low in nutrient elements, where organic matter has accumulated sufficiently in soils to produce the humic acids that give the river its dark colour and sufficient acidity to affect the plant and animal life it can support. The areas flooded in the annual cycle are forested and are known by the Portuguese word várzea. Trees in this zone survive flooding for several weeks annually and provide the basis of a food web that includes fish adapted for grazing on tree fruits and seeds. The grazing fish possess large flat molars adapted for masticating seeds and other coarse organic matter, and they compete for seeds dropped from pods in várzea trees on the river.
The distribution of the various zones of vegetation over the Earth has changed with climates throughout time. The greatest changes in the recent past have been due to the periods of glacial advance and retreat over the last several hundred thousand years. As recently as 15,000 years ago glacial ice covered much of eastern North America as far south as the present Hudson estuary and covered as well all of Scandinavia and northern Europe. Sea level was tens to hundreds of feet lower at various times, and extensive coastal areas now flooded were exposed. Such drastic changes in climate have sorted and resorted plant species, allowed the establishment of a diversity of forest types over time, and worked against the establishment or preservation of the mutual dependencies among species so common in the tropics. These higher-latitude forests, while similar in form and genera and sometimes in species around the world, are apparently highly mutable, the antithesis of the stable and self-perpetuating “organism” they were once thought to be. Nonetheless, the concept of the plant community as an organism remains viable and useful in ecology and is the basis of much of the most progressive analysis of how to manage the Earth for successful support of large numbers of people.
Succession and zonation
It is known from studies of plant residues and pollen preserved in the highly acid sediments of bogs and from observations of contemporary glaciers that the vegetation southward from the glacial front in the Northern Hemisphere was banded in much the same way the vegetation is zoned today: tundra occurred in a zone closest to the ice; coniferous forests occurred in a warmer and drier zone southward; and deciduous forests occurred still farther southward. As the habitat changed—that is, as the glacial ice melted and the glacial front retreated—the vegetation migrated onto the new landscape: first the pioneer species of the tundra—a few hardy low-growing or crustose lichens and mosses of small stature—followed by dwarfed willows and birch and, ultimately, the full panoply of tundra plants. As the climate ameliorated further, the forest followed, always according to pattern, with a few pioneers followed by the full array of species characteristic of the forest.
The process of invasion of a new landscape not previously occupied by plants has long been called “primary succession.” In this case the succession was in response to both the availability of a new habitat and a climatic warming that allowed the replacement of tundra by forest and of the coniferous forest ultimately by deciduous forest as the warming continued. Further warming might result in the replacement of deciduous forest by grassland, as has occurred worldwide at the steppe- or prairie-forest border in response to climatic changes over recent centuries. This boundary is strongly influenced by the fires that have swept grasslands throughout time. Disturbances such as clearings for agriculture, fires, diseases, and storms severe enough to open gaps in a forest may start secondary successional changes that also reestablish the normal vegetation for that climatic zone.
Throughout much of the tropics, where forests have been destroyed over large areas to make pasture, conditions appropriate for what might be a normal succession within disturbance-caused gaps in an otherwise intact forest do not exist, and the sites are permanently impoverished. The process generates grassland, shrublands, or sometimes bare earth. One-third of the land area of India has been impoverished in this way and is lost to agriculture, forests, or forestry. This class of land is the most rapidly growing class globally, according to interpretations of statistics of the United Nations Food and Agriculture Organization in 1985.
Ecosystems and the biosphere
The thin mantle of the Earth that supports life extends not more than a few feet into the sediments of the abyssal depths of the oceans and to a few tens of thousands of feet into the atmosphere, where pollen grains or other spores may survive. It is this thin layer at the surface of the Earth that is the biosphere, the place where life occurs. Human activity has changed the Earth irreversibly to form the present biosphere, and humans are now a force for further irreversible change.
Dispersal (dispersion) and colonization
The methods by which plants are distributed over the Earth are as diverse and complex as the plants themselves. The most widely occurring plants are the small-bodied, rapidly reproducing forms whose spores can be carried by wind and water and by birds and other animals.
Among the seed plants, whose spores are less mobile, explanations for the current distribution of plants become more complicated and include such profound changes over evolutionary time as the breakup of Pangaea some 300 million years ago, the opening of the Atlantic Ocean, and the isolation of North and South America, Australia, and Madagascar from larger continental landmasses. Progressive isolation produced endemism, evolutionary divergence sufficient to generate whole floras peculiar to a particular region, with many species, even genera, not known elsewhere. Volcanic islands are much younger than the continents and support floras derived from chance invaders carried by wind, sea, or animals, including humans. Island floras also come to exhibit endemism. Charles Darwin's observations of the fauna and flora of the Galápagos Islands (Galapagos Islands) off the western coast of South America led him to recognize the general process of biotic evolution. The islands are diverse in form and climate, isolated in varying degrees from each other and from the continent, and support a highly diversified flora and fauna clearly derived from South America, but modified over the time of isolation to contain forms peculiar to the islands.
Diverse mechanisms exist for dispersing spores and seeds at appropriate times and in appropriate habitats to ensure the survival of the new plant. In forests adapted to fire, pinecones may remain closed until a fire occurs. The heat of the fire opens the cones and releases the seeds into a fire-seared habitat, where the seedlings find soil with an abundance of nutrients left by the burning of organic matter and reduced competition from other plants. Seeds of trees frequently have “wings” that permit wider distribution by wind. Other plants, such as thistles and burdock, have awns or other appendages that catch on animals and obtain a wide distribution in areas frequented by them. Still others have elaborate mechanisms for spreading via rhizomes, stems, or other propagules.
Especially during the past two centuries, human activities have both deliberately and inadvertently spread the higher plants and microbial plagues of higher plants around the Earth. Examples among the higher plants are numerous; while many of the transfers of such plants have been benign and some clearly advantageous, others have been disastrous. Tree species have been moved freely around the world, sometimes with remarkable effectiveness. The Monterey pine (Pinus radiata), for instance, is a diminutive and unproductive tree on the coast of California, but it has become a major timber tree as a result of rapid growth in plantations in New Zealand. The rubber tree (Hevea) was carried in the 19th century from the moist tropics of Brazil to Java and elsewhere in the South Pacific, where its excellent growth, free of the diseases and competitors that affect it in its native American habitat, nearly destroyed the market for Brazilian rubber. Other inadvertent introductions have been far from benign; some have severely impoverished the landscape. Bromus tectorum, for example, is an annual grass introduced from Europe and the Transcaspian steppes to the arid intermontane west in North America, probably as a contaminant in fodder in the latter part of the 19th century. It spread rapidly and became a continuous ground cover over extensive areas. The plant completes its life cycle early in the season, sets seeds, and stands through the rest of the year as a continuous cover of dry grass not useful for fodder. It does, however, carry fire, which changes and can even destroy the native vegetation.
Microbial diseases of plants have been inadvertently spread around the world by human activities. Such diseases continue to exact a high cost in that they progressively impoverish the vegetation. One of the most profound changes in any forested region was produced by the introduction of the chestnut blight (Endothia parasitica) to North America. The fungus, introduced from Asia, found a home in the Fagaceae of eastern North America but was lethal to the American chestnut (Castanea dentata), the dominant species of extensive stands in the southern Appalachians and elsewhere. Once a common and popular shade tree and a source of smooth-grained, easily worked wood and of the abundant mast that was one of the principal foods of indigenous wild turkeys and other animals, the American chestnut never developed resistance to this now widely distributed fungus, which has several hosts in the Fagaceae and which produces wind-borne spores.
The American elm (Ulmus americana), a magnificent tree of the moist forests of lowlands throughout eastern North America, has suffered a similar but less comprehensive loss through the ravages of another exotic fungus, Dutch elm disease (Ceratocystis ulmi), which was imported from Europe on infected wood probably early in the 1900s. The fungus is spread by two elm bark beetles. Individual trees sometimes escape the fungus for many years but ultimately succumb before reaching the large sizes that were common throughout its range early in the 20th century.
Human effects on plants and natural communities
Humans have influenced the structure and development of natural communities for many thousands of years. The major influence has been through fire, which has been used deliberately in herding game, in rejuvenating plants used for fodder, in opening forested plots for agriculture, in maintaining savannas and grasslands, and in keeping forests open for easy passage for hunting. With the rapid expansion of human populations globally in recent decades, however, the changes in vegetation have been profound. Forests have advanced into grasslands and savannas. Succession has replaced pine forests with deciduous forests as in the southeastern coastal plain and piedmont regions of North America. In many instances, temporary protection from fire has resulted in the accumulation of a large mass of flammable vegetation that, when burning, makes a hot fire which kills the overstory trees and many species that normally survived the more frequent and less severe fires of the past. In such circumstances the vegetation may be impoverished, a condition that may prevail indefinitely, once established.
domestication
The origins of domesticated plants and agriculture (agricultural technology) are buried in a dim and unrecorded past of 10,000 years and more. Recent experience with Stone Age cultures of the Amazon basin and elsewhere has shown that such cultures have a sophisticated knowledge of plants for such purposes as food, medicine, and the tools and poisons used for hunting and fishing. Amerindian cultures improved a wide range of species by deliberate selection of the more productive and useful forms. Manioc (Manihot utilissima (cassava)) remains a staple of large sections of Latin America, especially Brazil and the Amazon basin. A woody, tuberous plant whose origin in the savannas of South America has long been lost, it is propagated vegetatively by planting a piece of tuber or a segment of stem. The tuber is ground to make a flour that must be washed to remove toxic quantities of hydrocyanic acid before being eaten directly or baked into a flat bread called cazabi (cassava).
corn, or maize (Zea mays), was domesticated 10,000 to 8,000 years ago either from teosinte (a perennial Zea that exists today) or from a lost ancestor that existed in the highlands of what is now central Mexico. Its culture had spread as far north as southern Maine by the time of European settlement of North America. Corn is now the third largest plant-based food source in the world. Other plants domesticated from the region include the common bean, squash, chili, tomato, avocado, papaya, guava, sapodilla, cotton, sisal, and vanilla.
The process of domestication appears to have involved selection and cultivation of the most promising and productive of plants long harvested in the wild. Continued selection brought drastic shifts in the population, shifts in the frequency of genes, and the development of new races totally dependent on humans to maintain them. Races developed in this way have been called cultigens to emphasize their dependence on cultivation. Such is the case with corn: the original stock has probably been lost, although teosinte may offer clues as to the source.
Major crop plants have been domesticated over the last several thousand years from sources identified by the Russian botanist N.I. Vavilov. Common wheat (Triticum vulgare) and rye (Secale cereale) probably were first domesticated from the grasses of Central Asia. Various millets (Panicum) and barley (Hordeum hexastichum) originated in the mountainous regions of central and western China, rice (Oryza sativa) probably in the Indian region.
Changes in biosystems: pollution
There is no plant community anywhere on Earth that has not felt the direct influence of the expansion of the human enterprise. The influence has had the form of direct intrusion through hunting and gathering, cultivation, or extensive harvest of trees or other plants or has had indirect effects through the harvest of fish or other animals or through changes in the chemistry of the environment. The latter changes are now pervasive and range from the introduction of pesticides, such as DDT, and other industrial toxins, such as the PCBs, to the acidification of rain, with substantial modification of the sulfur, nitrogen, and hydrogen ion budgets of ecosystems over large areas. Widespread destruction of forest tree species occurred during the 1980s in eastern North America and throughout Europe because of the combined effects of acid rain and other air pollutants.
Symptoms of incipient damage from air pollution and other chronic disturbances are now common in forests and other types of vegetation around the world, although symptoms may be subtle and difficult to assign as being caused by a particular pollutant. They range from such changes as the elimination of populations of leafy lichens from the bark of trees to the mortality of the trees themselves. Under extreme circumstances, such as in regions around smelters in Sudbury, Ont., Can., virtually all higher plants have been eliminated from an area of hundreds of square miles.
One of the most spectacular and informative examples of the patterns of impoverishment of forests exposed to chronic disturbance was induced experimentally in an oak-pine forest at Brookhaven National Laboratory in central Long Island, N.Y., U.S., using ionizing radiation. A single radiation source was used in the centre of the forest. The exposure, begun in the fall of 1961, was sufficient within months to eliminate all plants from a central area within a few metres of the source and to produce a systematic zonation of the forest at lower exposures. The zones ranged through a lichen and moss community, followed at lower exposures by a continuous ground cover of a single species of sedge (Carex pensylvanica). At slightly lower exposures a community of low shrubs occurred, then high shrubs at still lower exposures. An impoverished forest of hardy oaks well within the normal range of variability of the oak-pine forest of the region occurred down the gradient of exposure and beyond that zone; at exposures of one roentgen or less daily, the pines survived and the forest appeared intact and normal. Effects could, however, be detected by careful measurements of growth down to exposures on the order of 0.1 roentgen per day. The experiment offered a specific example of the progressive impoverishment of vegetation where both cause and effect could be measured, something not possible in most instances of pollution. Such experience is useful not only in defining the effects of exposure to ionizing radiation but also in identifying stages in the impoverishment of natural communities as effects accumulate.
Similar changes occur in aquatic systems in response to chronic pollution. The early stages of pollution in bodies of water usually involve enrichment with nutrient elements, especially nitrogen and phosphorus, a process known as eutrophication.
The effects of pollution on land and in water are to favour small-bodied, rapidly reproducing organisms that do not depend on complex food webs. The process of simplification and impoverishment is now global and affects terrestrial and aquatic communities alike. It is the continuously expanding result of chronic intrusions on natural systems by human influences. The impoverishment threatens all life because it reduces systematically the capacity of the Earth to support plants.
conservation
One of the most serious threats to life is the possibility of a rapid warming (global warming) of the Earth from the accumulation of heat-trapping gases in the atmosphere. The most important of the gases is carbon dioxide, but methane, nitrous oxide, and the chlorofluorocarbon compounds (CFCs) also contribute significantly. The largest sources of carbon dioxide are the combustion of fossil fuels, the destruction of forests, and, probably, the warming itself. methane is a product of the use of fossil fuels and of the decay of organic matter in soils and swamps and bogs. Its rate of evolution is accelerated by warming.
The effects of the warming are expected to be profound: If the warming occurs as rapidly as some suggest, there could be a change in the average temperature of the Earth of as much as 0.5°–1° C (0.9°–1.8° F) per decade during the early part of the 21st century. A change of 1° in temperature is equivalent to 60–100 miles of latitudinal change in the middle latitudes. Such high rates of change in climatic zones would not only disrupt agriculture but also destroy forests and other natural communities at their warmer and drier margins more rapidly than they can be regenerated elsewhere. One of the effects would be to speed the decay of organic matter in soils and accelerate the accumulation of carbon dioxide and methane in the atmosphere, an effect that would further accelerate the warming. Avoiding such change will require abandoning fossil fuels (fossil fuel) as the major source of energy for industrial societies. An alternative is solar energy.
Classical approaches to conservation involve specific efforts at preserving species that are endangered. By far the most desirable approach to the preservation of the Earth's 5–30 million or more species is through management of natural ecosystems. Studies of tropical forests have emphasized that the area required to assure the habitat of bird populations as well as populations of other animals and plants is very large indeed, on the order of 10,000 hectares. There is evidence to support earlier experience that the best way to assure the preservation of the indigenous biota is to maintain a matrix of the indigenous vegetation with more than 50 percent of the land area undisturbed and an additional 25 to 50 percent in successional stages. This type of land-use planning exists in very few places around the world but offers the advantage of a landscape that maintains itself with stable, near-maximum flows of energy through living systems and offers, regionally, maximum sustainable support for people at low cost. The difficulty is that the apparently high stocks of land under light use—the forests, the water, and energy in various forms—are an open invitation to immediate, short-term exploitation, and their existence may suggest that the landscape can be more intensively occupied.
evolution and paleobotany
The evolutionary history of plants is recorded in fossils preserved in lowland or marine sediments. Some fossils preserve the external form of plant parts, others show cellular features, and still others consist of microfossils such as pollen and spores. In rare instances, fossils may even display the ultrastructural or chemical features of the plants they represent. The fossil record reveals a pattern of accelerating rates of evolution coupled with increasing diversity and complexity of biological communities that began with the invasion of land and continued with the progressive colonization of the continents. At present, fossil evidence of land plants dates to the Ordovician Period (505 to 438 million years ago) of the Paleozoic Era.
By far the most diverse and conspicuous living members of the plant kingdom are vascular plants (tracheophytes (tracheophyte)), in which the sporophyte phase of the life history (see above Reproduction and life histories: Life histories (plant)) is dominant. Fossil remains of vascular plants provide evidence for evolutionary changes in the structure of the plant body (sporophyte and gametophyte), in the variety of plant forms, in the complexity of the life history, in the tolerance for ecological conditions, and in systematic diversity. Nonvascular plants, or bryophytes (bryophyte) (mosses, liverworts, and hornworts), are much smaller and less diverse than vascular plants. The first evidence for liverworts is from rocks of the Devonian Period (408 to 360 million years ago), while the earliest moss fossils are from the Permian Period (286 to 245 million years ago). In contrast to tracheophytes, most fossil bryophytes are relatively similar to living forms. Understanding of the evolution of nonvascular plants is, therefore, less complete than for tracheophytes.
Evolution of land plants in the Ordovician through Middle Devonian periods
Botanists now believe that plants evolved from the algae; (algae) the development of the plant kingdom may have resulted from evolutionary changes that occurred when photosynthetic multicellular organisms invaded the continents. The earliest evidence for land plants consists of isolated spores, tracheid-like tubes, and sheets of cells found in Ordovician (Ordovician Period) rocks. The abundance and diversity of these fossils increases into the Silurian Period (438 to 408 million years ago), where the first macroscopic (megafossil) evidence for land plants has been found. These megafossils consist of slender forking axes that are only a few centimetres long. Some of the axes terminate in sporangia that bear trilete spores (i.e., spores that divide meiotically to form a tetrad). Since a trilete mark indicates that the spores are the product of meiosis, the fertile axes may be interpreted as the sporophyte phase of the life cycle.
Fossils of this type could represent either vascular plants or bryophytes. Another possibility is that they are neither but include ancestors of vascular plants, bryophytes, or both. The earliest fossils also include at least one or more additional plant groups that became extinct early in the colonization of the land and therefore have no living descendants. By the Early Devonian Period (408 to 360 million years ago), some of the fossils that consist of forking axes with terminal sporangia also produced a central strand of tracheids, the specialized water-conducting cells of the xylem. Tracheids are a diagnostic feature of vascular plants and are the basis for the division name, Tracheophyta.
The simplest and presumably most primitive vascular plants from the Late Silurian and Early Devonian periods (Devonian Period) (421 to 387 million years ago) were the Rhyniopsida. They included plants such as Cooksonia and Rhynia, which were herbaceous colonizers of moist habitats. Most were less than 30 centimetres (12 inches) tall. The plant body was not differentiated into stems, leaves, and roots; rather, the forking, above-ground axes bore terminal sporangia and produced stomata which demonstrate that the plants were green and photosynthetic. Surface or underground axes served to root the plant and were anchored by rhizoids. Because such plants produced only one type of spore, they were nonseed plants with a homosporous life cycle and free-living gametophytes. A small number of such gametophytes have been described from Devonian deposits.
Some plants of the Early Devonian had multicellular emergences of tissue along their above-ground axes, which are thought to have increased the light-capturing surface of the photosynthetic tissue. Such emergences (enations) gave rise to the leaves (leaf) (microphylls) of the Lycopsida, thus producing an above-ground shoot system that consisted of branching stems with leaves. Underground axes that lacked leaves would have become the roots. Lycophytes were the first plants with well-differentiated shoot systems, and they are the most ancient groups of vascular plants with living representatives.
The leaves of several other plant groups were derived from modifications of the forking axes. There was a variety in structure among Devonian plants. The axes of some plants forked equally, while other plants—e.g., Drepanophycus—were more specialized, comprising a large, centrally located axis as well as smaller axes borne laterally. In plants where the lateral systems branched in only one plane, the side branches were flat like leaves. Filling in (webbing) of the spaces between forks of the laterals with photosynthetic tissue produced leaves called megaphylls. There is evidence for the evolution of megaphylls in several plant groups of the Late Devonian Period (374 to 360 million years ago) and Early Carboniferous Period (360 to 320 million years ago). Although most of these groups have no living representatives, by the Carboniferous Period they had given rise to homosporous ferns, sphenopsids (horsetails), and seed plants (gymnosperms).
As Devonian plants with microphylls and those with specialized branching systems diversified, many grew to the size of shrubs. By the Middle Devonian Period (387 to 374 million years ago) there were shrub-size representatives of several lineages, but a further increase in the size of herbaceous plants was restricted by the limited diameter that above-ground stems and rooting systems could achieve. The development of lateral (secondary) growth overcame this size restriction. The ability to produce secondary growth evolved independently in several groups. In the lycophytes, much of this secondary growth occurred in cortical tissues; in the ancestors of seed plants and several other lineages, however, the production of wood accounted for most of the growth in stem diameter. About the same time, downward-growing, central rooting (root) systems evolved independently in lycophytes and other plant groups. As a result there were forests with a canopy of giant lycophyte and gymnosperm trees near the beginning of the Early Carboniferous Period.
As plants developed more complex growth forms, they also underwent systematic diversification and evolved more specialized modes of sexual reproduction. The most primitive vascular plants had a homosporous life cycle, in which reproduction and dispersal involved a single type of spore. Extant homosporous plants include most ferns and many lycophytes. The homosporous life cycle is an effective means for long-distance dispersal of species. Although it permits the fertilization of an egg by a sperm from the same gametophyte plant, genetic recombination, considered important for more rapid evolution, is not possible. Moreover, because the gametophytes of homosporous plants are exposed to the environment for an extended period of time, mortality is relatively high.
By the Middle Devonian Period (387 to 374 million years ago), the heterosporous life cycle had evolved independently in several groups, including lycophytes and the ancestors of seed plants. In heterosporous plants there are two sizes of spores; the smaller (a microspore) produces a male gametophyte, and the larger (a megaspore) produces a female gametophyte. The incidence of genetic recombination is increased by this obligate cross-fertilization. Both types of gametophytes develop quickly within the protective spore wall. Compared with homosporous plants, reproduction is more rapid and mortality is reduced in heterosporous plants. By the end of the Devonian, heterosporous plants became the dominant species in most wetland environments; however, the need for an abundant source of water from the environment to effect fertilization prevented the heterosporous plants from establishing communities in drier habitats.
Evolution of seed plants and plant communities
A series of changes in the reproductive biology among some heterosporous plants during the Late Devonian overcame this environmental restriction and allowed them to colonize a much wider range of habitats. These changes also led to the evolution of seed plants. In seed plants, the megaspore is retained in the megasporangium and the microspore is brought to a pollen chamber at the tip of this organ. The megasporangium is then sealed, and gametophyte development and fertilization occur within a protected environment. When such a megasporangium is enclosed in a seed (seed and fruit) coat, the structure with its enclosed embryo is called a seed. Seeds of the earliest such plants were exposed to the external environment; these “naked seed” plants are referred to as gymnosperms (gymnosperm).
From the Late Devonian through the base of the Late Cretaceous Period (97.5 to 66.4 million years ago), gymnosperms underwent dramatic evolutionary radiations and became the dominant group of vascular plants in most habitats. Extant gymnosperms include conifers, cycads, and Ginkgo biloba, but these represent only a small fraction of the gymnosperms that inhabited the Earth during the Mesozoic Era (245 to 66.4 million years ago). Among the Mesozoic forms were species with a wide variety of mechanisms for effecting pollination, protecting the seeds, dispersing the seeds, and increasing the natural selection of the most successful varieties.
Primitive forms of the flowering plants (angiosperms (angiosperm)) arose from among this diverse array of complex gymnosperms. From their earliest diversification in the Cretaceous Period (144 to 66.4 million years ago), angiosperms rapidly came to dominate the land flora. Today there are approximately 250,000 to 300,000 species of flowering plants, which account for more than 90 percent of the diversity of vascular plants. Among the many structures that contribute to this success are flowers, fruits, complex vein patterns in the leaves, and highly specialized cells of the conducting system.
Just as an ecological succession of plant forms can transform bare ground into a complex plant community, so an evolutionary succession can be thought of as having transformed bare continents into rich terrestrial biotas. In ecological succession, herbaceous weeds colonize bare earth and modify the environment for the development of more complete ground cover. This further modifies the environment for the successive establishment of larger herbs, perennial shrubs, fast-growing trees, and, finally, slower-growing trees, vines, and epiphytes (plants that grow on other plants rather than in soil).
As discussed above, primitive land plants of the Late Silurian and Early Devonian periods were primarily small herbs that inhabited the moist lowlands near oceans, lakes, and streams. The first of these grew on bare ground because there would have been little or no organic soil. They may be thought of as the evolutionary equivalent of primary colonizing weeds, and their establishment on land was responsible for the first of three dramatic increases in the diversity of land plants.
After the initial production of organic soils by these evolutionary colonizers, communities of larger herbs and shrubs were able to develop, and they became common in the Middle Devonian. In the Late Devonian these communities were themselves succeeded by communities dominated by heterosporous, tree-sized plants. During the Early Carboniferous Period, nonseed plants continued to dominate many wetland habitats, while communities dominated by gymnosperm trees colonized drier habitats than had been previously available to the heterosporous forms.
Communities dominated by trees were the first to provide suitable habitats for vines and for epiphytes. Such communities also provided habitats for herbivorous animals. As a result, biological communities of considerable complexity evolved by the Late Carboniferous Period (320 to 286 million years ago), giving rise to a second dramatic increase in the diversity of land plants.
With the rapid diversification of the gymnosperms and an increasing complexity of interactions between plants and animals, there was a significant evolutionary turnover among land plants during the Permian, Triassic, and Jurassic periods (286 to 144 million years ago). New groups of gymnosperms replaced some of the primitive gymnosperms as well as the most prominent nonseed plants from earlier periods. Among the most successful gymnosperms of these periods were conifers, cycads, Ginkgo, and several major groups with no extant representatives.
Flowering plants probably also originated during this time but did not become a significant part of the fossil flora until the middle of the Cretaceous Period. The fossil evidence provides a clear picture of the rapid diversification and spectacular rise to dominance of the angiosperms during the Late Cretaceous Period. This was accompanied by a dramatic increase in the complexity of plant community structure, produced at least in part by an expanding array of interactions between plants and animals. Improved pollination and seed dispersal were among the benefits of such interactions to plants. That animals equally benefited is evidenced by the coevolution of several groups of animals (particularly insects) and angiosperms during the Late Cretaceous and Tertiary periods. Such interactions contributed significantly to a third rapid increase in global plant diversity and helped angiosperms achieve the overwhelming dominance of the land flora characteristic of modern vegetation.
Classification
Annotated classification
Kingdom Plantae
Multicellular, photosynthetic, eukaryotic organisms containing chlorophylls a and b and carotenoids and forming true starch; chlorophyll contained in stacks of membranes (grana) within chloroplasts; cellulosic cell walls; aerial tissues covered with a waxy cuticle and provided with openings (stomata) in the epidermis bordered by two guard cells and serving in gas exchange; alternation of generations with multicellular gametangia and sporangia and possessing a multicellular embryo stage; motile sperm, when present, with whiplash flagellum; sperm with a microtubular cytoskeleton; cell division associated with the dispersal of the nuclear membrane and the formation of a cell plate across the mitotic spindle (phragmoplast).
Division Bryophyta (bryophyte)
Small, mostly nonvascular, archegoniate plants with a dominant, photosynthetic, free-living gametophyte; sporophyte has little or no chlorophyll and is dependent on gametophyte; biflagellate sperm.
Class Musci (mosses (moss))
Gametophytes “leafy” and radially symmetrical, with leaves arising spirally from stemlike axis; leaves rarely notched or lobed, with thickened “midrib”; many chloroplasts per leaf cell; gametophyte with multicellular rhizoids; sporophytes with complex spore-containing capsule with peristome and operculum, columella, and stomata present, but elaters absent; between 10,000 and 14,000 species; representative genera include Polytrichum (hair-cap moss), Mnium, Funaria (cord moss), and Sphagnum.
Class Hepaticae (liverworts (liverwort))
Gametophytes either “leafy” or dorsiventrally flattened (strap-shaped) and thalloid; leafy forms with leaves in 3 rows, 2 lateral and 1 below; leaves usually notched or lobed, with thickened midrib lacking; gametophytes with unicellular rhizoids; many chloroplasts per cell; sporophytes ephemeral, surrounded by gametophytic tissue, lacking opercula, peristome, stomata, and columella; elaters mixed with spores in capsule; capsule opening into 4 or more valves; between 6,000 and 9,000 species; representative genera include Porella, Frullania, Marchantia, Conocephalum, and Riccia.
Class Anthocerotae (hornworts (hornwort))
Gametophyte thalloid, with a single, large chloroplast per cell, mucilage cavities present; sporophytes persistent, erect (hornlike), photosynthetic, arising from upper surface of gametophyte, possessing stomata, columella, basal meristem, and pseudoelaters opening by 2 valves but lacking an operculum; representative genus, Anthoceros.
Division Lycophyta (lycophyte) (club mosses, spike mosses, quillworts)
Vascular plants; sporophyte differentiated into stem, leaf, and root; leaves spirally arranged on stem, of the microphyll type; sporangia borne on upper (adaxial) surface of leaves (sporophylls), one sporangium per sporophyll; vascular tissue basically forming a central core in stem (protostelic); homosporous or heterosporous; ligulate (having basal, leaflike protuberances) or eligulate; strobili (cones) present or lacking; all living genera with primary growth only; gametophytes subterranean or surface-dwelling; motile sperm; about 5 living genera and between 600 and 1,000 species; representative genera include Lycopodium (club moss), Selaginella (spike moss), and Isoetes (quillwort).
Division Psilotophyta (whisk ferns)
Vascular plants; sporophyte lacking roots and often leaves; stems with small enations, dichotomously branched; vascular tissue forming a central core in stem (protostelic); sporangia fused into synangiate structure, apparently terminal on short stem; homosporous; gametophytes subterranean, with motile sperm; representative genus, Psilotum (whisk fern).
Division Sphenophyta (Equisetopsida) (horsetails, scouring rushes)
Vascular plants; sporophyte differentiated into stem, leaf, and root; stems ribbed and jointed, monopodial; minute leaves whorled at the nodes; vascular tissue organized into bundles; sole living genus with primary growth only; sporangia borne on specialized stalks (sporangiophores) in strobili; homosporous; gametophytes photosynthetic, surface-dwelling; motile sperm; 1 genus, Equisetum (horsetail).
Division Filicophyta (ferns (fern))
Vascular plants; sporophyte differentiated into stem (rhizome), roots, and leaves (fronds); leaves entire or, more often, divided; arrangement of vascular tissue in stem variable, primary growth only; sporangia usually clustered into sori, often located on the under (abaxial) surface of sporophylls; mostly homosporous; gametophytes (prothallia) either subterranean and nongreen or, more commonly, surface-dwelling and photosynthetic; sperm motile; between 9,000 and 12,000 species; representative genera include Pteridium, Polypodium, Polystichum, Adiantum, and Cyathea.
Division Cycadophyta (cycadophyte) (cycads)
Palmlike, gymnospermous plants with typically short, thick, unbranched aerial trunks, sometimes subterranean, and large, divided leaves; leaves usually thick and leathery; cones present, often large and terminal on the stem; pollen (male) and seed (female) cones borne on separate plants; gametophytes reduced, not free-living; sperm motile (flagellated), but brought to vicinity of egg by a pollen tube; 10 genera and about 120 species; representative genera include Zamia and Cycas.
Division Ginkgophyta (ginkgophyte) (ginkgoes)
Gymnospermous plants; tall, much-branched tree with well-developed cylinder of wood; resin ducts present; xylem with tracheids only; stem differentiated into long shoots and short spur shoots; simple, fan-shaped leaves with open dichotomous venation terminate short shoots; leaves deciduous; sexes on separate trees; distinct cones lacking; gametophytes reduced, not free-living; sperm motile (flagellated); mature seeds with fleshy, foul-smelling outer region; 1 living species, Ginkgo biloba.
Division Coniferophyta (conifer) (conifers)
Gymnospermous plants; mostly trees with abundant xylem composed of tracheids only; resin ducts present; leaves simple, needlelike, scalelike, with a single vein or, less commonly, strap-shaped with multiple veins; reproduction by well-defined cones; seeds exposed on ovuliferous scales; gametophyte generation reduced, microscopic, not free-living; sperm nonmotile, transported to egg by pollen tube; approximately 50 to 55 genera and between 550 and 575 species; representative genera include Pinus (pine), Abies (fir), Sequoia, Taxodium, Juniperus (juniper), Cupressus (cypress), and Agathis.
Division Gnetophyta (gnetophyte) (gnetophytes)
Diverse and unusual group of 3 gymnospermous genera; growth habits of all 3 are atypical among gymnosperms in possessing vessel elements in the xylem and reproductive structures that are somewhat flowerlike; gametophytes reduced as in other gymnosperms; sperm nonmotile; extant genera Gnetum, Ephedra, and Welwitschia.
Division Magnoliophyta (flowering plants, angiosperms (angiosperm))
Vascular plants, xylem typically with vessel elements; reproduction by flowers; ovules or young seeds enclosed by female reproductive structure (carpel); gametophyte generation extremely reduced, consisting of only a few cells; archegonia and antheridia lacking; nonmotile sperm transported to egg by pollen tube; pollen transported to specialized receptive surface (stigma) on carpel; double fertilization, one sperm uniting with the egg to form a zygote, another fusing with the polar nuclei to form the primary endosperm nucleus; nutritive tissue of seed triploid endosperm; seeds enclosed by mature ovary that ripens into a fruit; approximately 230,000 species worldwide, more than 300 families.
Class Magnoliopsida (Dicotyledonae (dicotyledon); dicots)
Angiosperms with 2 seedling leaves (cotyledons); leaves with reticulate (net) venation; stem with vascular bundles arranged in a ring, each bundle with a functional vascular cambium; secondary growth common; flower parts in multiples of 4 or 5; pollen typically tricolpate (3 germinal apertures) or some derived type; about 165,000 species.
Class Liliopsida (Monocotyledonae; monocots)
Angiosperms with 1 seedling leaf (cotyledon); leaves with parallel venation; stem with scattered vascular bundles; bundles without a functional vascular cambium; secondary growth typically absent; flower parts in multiples of 3; pollen typically have a single germinal aperture; approximately 50,000 to 55,000 species; representative types include grasses, orchids, palms, and lilies.
Critical appraisal
In the classification above, only the major divisions and classes of living plants are listed, and a number of entirely extinct divisions are omitted. The classification outlined is somewhat conservative but is one that best conforms to available data and has gained wide acceptance.
Biological classifications were initially mechanical or “artificial”; that is to say, they had no basis in evolution. This was followed by a period of “natural system” construction, whereby plants were grouped together on the basis of their overall similarities or differences, using as many characteristics as possible. Contemporary systems of biological classification are phylogenetic, which means that various plants are arranged together because they are thought to be related by descent from a common ancestor. As more evidence is accumulated, it is to be expected that classifications also will change to accommodate this new information.
At the turn of the 19th century, the plant kingdom was frequently divided into two major groups, the cryptogamia (algae, fungi, bryophytes, and ferns) and the phanerogamia (gymnosperms and angiosperms). Subsequently, it was common practice among systematic botanists to group all vascular plants together under a single division, Tracheophyta. In contrast, all of the major individual groups of plants are now most often elevated to divisional rank, a view that interprets the individual major groups to be less closely related to one another than previously believed. Difficult and complex questions still exist in the definition and circumscription of certain groups. Although it is convenient to treat the Bryophyta (mosses, liverworts, and hornworts) as a single division, some authorities prefer to recognize three or five separate divisions for these plants, each representing an independent evolutionary lineage. Furthermore, the phylogenetic relationships, if any, of the bryophytic plants with primitive vascular plants remains unclear. The treatment of the Psilotophyta is controversial. Although here recognized as a separate division of lower vascular plants, some botanists prefer to include the group among the ferns (Filicophyta). The flowering plants have classically been recognized as composed of two distinct classes; however, the origin of each class and the phylogenetic relationships of their component taxa have yet to be satisfactorily resolved.
Additional Reading
General surveys providing definitions of the kingdom and characterizations of major groups of plants include Peter H. Raven, Ray F. Evert, and Susan E. Eichhorn, Biology of Plants, 4th ed. (1986), an excellent introductory text; Harold C. Bold, Constantine J. Alexopoulos, and Theodore Delevoryas, Morphology of Plants and Fungi, 5th ed. (1987), a detailed and comprehensive treatment that includes algae and fungi; Ernest M. Gifford and Adriance S. Foster, Morphology and Evolution of Vascular Plants, 3rd ed. (1989), an advanced text; and V.H. Heywood et al. (eds.), Flowering Plants of the World (1978, reissued 1985), a beautifully illustrated guide. Henry S. Conard, How to Know the Mosses and Liverworts, 2nd ed., rev. by Paul L. Redfearn, Jr. (1979); and W.B. Schofield, Introduction to Bryology (1985), are well-illustrated guides to the bryophytes. John T. Mickel, How to Know the Ferns and Fern Allies (1979), is an introduction to the ferns and lower vascular plants of the United States.Plant physiology is the subject of many specialized studies. T. Wallace, Trace Elements in Plant Physiology (1950), is the classic exposition of the pioneering experimentation on and founding principles of the subject. Emanuel Epstein, Mineral Nutrition of Plants: Principles and Perspectives (1972), treats the subject from the perspective of the basic and applied plant sciences; John Skujins, “Essential Nutrient Elements and Nutrient Cycles in Soil,” pp. 41–48 in vol. 3 of David W. Newman and Kenneth G. Wilson (eds.), Models in Plant Physiology and Biochemistry, 3 vol. (1987), offers a concise overview of the subject; A.D.M. Glass, Plant Nutrition: An Introduction to Current Concepts (1989), provides a complete exposition suitable for the informed general reader; and A. Läuchli and R.L. Bieleski (eds.), Inorganic Plant Nutrition (1983), is a definitive advanced treatment of mineral nutrition. Metabolic cycles and photosynthesis in plants are studied in Cecie Starr and Ralph Taggart, Biology: The Unity and Diversity of Life, 5th ed. (1989); David D. Davies (ed.), Metabolism and Respiration (1980), Biochemistry and Metabolism (1987), and Physiology of Metabolism (1987); G. Ray Noggle and George J. Fritz, Introductory Plant Physiology, 2nd ed. (1983), which provides an overview of plant metabolism; and Frank B. Salisbury and Cleon W. Ross, Plant Physiology, 3rd ed. (1985), which offers a more advanced treatment. Annual Review of Plant Physiology and Plant Molecular Biology covers current research in all subjects of plant physiology.Ecological considerations are discussed in Michael G. Barbour, Jack H. Burk, and Wanna D. Pitts, Terrestrial Plant Ecology, 2nd ed. (1987); Victor V. Rendig and Howard M. Taylor, Principles of Soil-Plant Interrelationships (1989); Friedrich G. Barth, Insects and Flowers: The Biology of a Partnership (1985; originally published in German, 1982); and Oswald Tippo and William Louis Stern, Humanistic Botany (1977), on the impact of plants on human beings. On the cultivation of plants, see N.I. Vavilov, The Origin, Variation, Immunity, and Breeding of Cultivated Plants: Selected Writings, trans. from Russian (1951); and R. Walden, Genetic Transformation in Plants (1989).Analyses of evolutionary relationships are provided in Barry A. Thomas, The Evolution of Plants and Flowers (1981), a spectacularly illustrated introduction; William G. Chaloner, “Early Land Plants: The Saga of a Great Conquest,” pp. 301–316 in Werner Greuter and Brigitte Zimmer (eds.), Proceedings of the XIV International Botanical Congress (1988), a summary of what is known about the earliest land plants; Wilson N. Stewart, Paleobotany and the Evolution of Plants (1983), on the fossil history and the evolution of plant structures; and Else Marie Friis, William G. Chaloner, and Peter R. Crane (eds.), The Origins of Angiosperms and Their Biological Consequences (1987), a collection of writings on the geography, climate, ecology, plant-animal interactions, structure, reproductive biology, and interrelationships of flowering plants in their evolution from the Cretaceous to the modern biota.For classification, see Lynn Margulis and Karlene V. Schwartz, Five Kingdoms: An Illustrated Guide to the Phyla of Life on Earth, 2nd ed. (1988), a brief account of the diversity and important traits of the major groups of living organisms according to the five-kingdom system of classification; Albert E. Radford et al., Vascular Plant Systematics (1974), a comprehensive sourcebook on all areas of higher plant systematics and a complete survey of terminology; and Arthur Cronquist, An Integrated System of Classification of Flowering Plants (1981), a scholarly outline of the modern system of angiosperm classification based on available information.
- Denmark Strait
- Denmark Vesey
- Denner, Johann Christoph
- Dennett, Mary Coffin Ware
- Dennie, Joseph
- Dennis
- Dennis Brutus
- Dennis C. Blair
- Dennis, Eugene
- Dennis Gabor
- Dennis, John
- Dennis Kucinich
- Dennis, Nigel
- Dennison, Aaron Lufkin
- Dennis Robert Hoagland
- Dennis the Menace
- Dennstaedtiaceae
- Denon, Dominique Vivant, Baron
- Denpasar
- densitometer
- density
- Density changes with depth (seawater 35 parts per thousand and 0 C)
- density current
- Density values of seawater*
- Densmore, Frances